baabyylewis
16.03.2020 •
Biology
101. Du
An insulated Cup contains 75.0 g of water
at 24.00 C. A 26.00 Sample of metal at
82.
25oC is added. The finas temperture of the
water and metal is 28.
34 C. What is the
specific heat of the metal?
Solved
Show answers
More tips
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- F Family and Home What is Most Important in Men s Lives?...
- G Goods and services Which TV is better - LCD or Plasma?...
- C Computers and Internet Are there special gaming mice?...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
- C Computers and Internet 3D Glasses! What is this thing?...
Answers on questions: Biology
- M Mathematics Find the area of this triangle. Round to the nearest tenth. 8m, 62°, 12m....
- G Geography Match the climate change term in column 1 to the characteristic in column 2. Human activity ? Produces greenhouse gases Sun s output ? Peaked 20,000 years ago Ice age ? Can vary...
- M Mathematics im putting this up and ill look at it tmrw im trying to help a friend end things with her abusive bf any other ideas besides calling the police or leave cuz she cant do that thanks...
- B Biology Anurse is caring for a client with chronic kidney failure. which clinical findings should the nurse expect when assessing this client? select all that apply....

Ответ:
Explanation:
When the sample of hot metal is placed inside the water, the metal gives off heat to the water: the temperature of the metal decreases while the temperature of the water increases, until the two reach an equilibrium temperature.
The heat given off by the metal is equal (in magnitude) to the heat absorbed by the water, so we can write:
where:
And solving for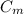 , we find the specific heat of the metal:
, we find the specific heat of the metal:
Ответ:
you to
Explanation: