Amixture of oxygen, hydrogen, and nitrogen exerts a total pressure of 378 kpa. if the partial pressures of oxygen and hydrogen are 212 kpa and 101 kpa respectively, find the partial pressure exerted by nitrogen.
a. 65 kpa
b. 313 kpa
c. 166 kpa
d. 277 kpa
Solved
Show answers
More tips
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Biology
- M Mathematics Complete parts (a) through (c) below. a. A storage pod has a rectangular floor that measures 20 feet by 10 feet and a flat ceiling that is 6 feet above the floor. Find the area of...
- H Health A28-year-old man presents with reports of diarrhea, fecal urgency, and weight loss. his stool is light-colored and malodorous, and it tends to float and be difficult to flush. he has...
- E English I used also to carry bread with me, enough of which was always in the house, and to which I was always welcome; I was much better off in this regard than many of the poor white children...
- B Business John is over seven feet tall. When asked whether John is a professional basketball player or a software programmer, many people predict the former, even though there are many more...

Ответ:
As we know that
P total = pO +pH +pN
so
P = P1 + P2 + P3
378 kPa= 212 kPa + 101 kPa + P(H2)
P(H2) = 65 kPa
partial pressure is 65
so correct option is A
hope it helps
Ответ:
The correct answer is option A.
Explanation:
Total pressure of mixture = p = 378 kPa
Partial pressure of the oxygen gas =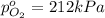
Partial pressure of the hydrogen gas =
Partial pressure of the nitrogen gas =
As we know that total pressure is sum of partial pressures exerted by all the individual gases.
65 kPa the partial pressure exerted by nitrogen.
Ответ:
C
Explanation:
Operating leverage may be defined as the potential use of fixed operating costs to magnify the effects of changes in sales on a firm's earnings before interest and taxes