crystalclear99
21.03.2020 •
Chemistry
0.100 mol of CaCO3 and 0.100 mol CaO are placed in an 10.0 L evacuated container and heated to 385 K. When equilibrium is reached the pressure of CO2 is 0.220 atm. 0.300 atm of CO2 is added, while keeping the temperature constant and the system is allowed to reach again equilibrium. What will be the final mass of CaCO3
Solved
Show answers
More tips
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- L Leisure and Entertainment What can be considered an antique: defining and valuing old objects...
- L Leisure and Entertainment Choosing the Right Books to Read: Tips and Recommendations...
- C Computers and Internet Step-by-Step Guide on How to Download Music to Your iPhone...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- F Family and Home How to Choose Suitable Windows for Your Home?...
- L Leisure and Entertainment What Movies You Should Watch: A Guideline to Make the Right Decision...
- L Leisure and Entertainment How to Identify a Song? Which Program Can Recognize a Song by Its Melody?...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- F Family and Home Parquet or laminate, which is better?...
Answers on questions: Chemistry
- C Chemistry The molar mass of h2o is 18.015 g/lol. how many grams of h2o are present in 0.20 mol...
- C Chemistry What type of climate is found in the tundra?...
- C Chemistry Please can you help me with this...
- C Chemistry During which of the following processes does a glucose molecule break down into ethanol? Aerobic respiration Alcoholic fermentation Lactic acid fermentation Photosynthesis...
- C Chemistry How many electrons will oxygen give up to become stable...
- C Chemistry Which statement describes all solids? They contain loosely packed atoms. They have a definite shape and volume. They have a smooth, rigid surface. They flow with resistance....
- C Chemistry What is the pH of a solution that has a pOH of 5.4?...
- C Chemistry Which molecule has ionic bond? Mg C0 NaCl N0...
- C Chemistry Which image represents ether? Check all that apply. A skeletal model has 6 carbons, with an oxygen between the first and second carbon. A 5-carbon chain, with O H before...
- C Chemistry 1. Nhiệt dung của một chất là gì? Tính nhiệt lượng cần thiết để nâng nhiệt độ của 200 gam nước từ -200C đến 2000C ở áp suất 1 atm. Biết rằng: Nhiệt nóng chảy của nước ở...

Ответ:
12.531 grams will be the final mass of calcium carbonate.
Explanation:
Partial pressure of carbon dioxide gas at equilibrium =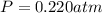
Volume of carbon dioxide gas = V = 10.0 L
Moles of carbon dioxide gas formed = n
Temperature of the gas = T = 385 K
According to reaction , 1 mole of carbon dioxide gas is formed from 1 mole of calcium carbonate,0.06960 mole of carbon dioxide gas will be obtained from :
Moles of calcium carbonate at equilibrium = 0.100 mol - 0.06960 mol = 0.03040 mol
After addition of 0.300 atm of carbon dioxide gas, more amount of calcium carbonate will be be formed.
Here, at the same temperature, the equilibrium pressure of the carbon dioxide gas is 0.220 atm so, entire 0.300 atm of carbon dioxide will get convert to calcium carbonate.
So amount moles of carbon dioxide gas added = moles of calcium carbonate formed after re-establishment of an equilibrium :
Partial pressure of carbon dioxide gas added at equilibrium =
Volume of carbon dioxide gas = V = 10.0 L
Moles of carbon dioxide gas formed = n
Temperature of the gas = T = 385 K
Moles of calcium carbonate = 0.09491 mol
Total moles of calcium carbonate in the container :
=0.03040 mol + 0.09491 mol = 0.12531 mol
Mass of 0.12531 moles of calcium carbonate :
0.12531 mol × 100 g/mol = 12.531 g
12.531 grams will be the final mass of calcium carbonate.
Ответ:
i dont have tictok im only 11
Explanation: