britishgirl1
19.05.2020 •
Chemistry
1.What mass of oxygen gas,O2, from the air is consumed in the combustion of 702g of octane, C8H18, one of the principal components of gasoline? 2C8H18 + 25O2 -> 16CO2 + 18H2O.
Solved
Show answers
More tips
- F Family and Home When and how to start introducing solid foods to your baby?...
- B Business and Finance Moneybookers – What it is and How it Works...
- C Computers and Internet How to Format Your C Drive: Detailed Guide and Tips...
- F Food and Cooking What can and cannot be eaten during Lent?...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- D Dating, Love, Relationships Is it a Compliment or Flattery: What s the Difference?...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- C Computers and Internet What to Do If Your ICQ Gets Hacked?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry Sample of acid are brought to a laboratory for analysis. several titrations are performed and it is determined that a 20.0-mililiter sample of acid rain is neutralized with 6.50...
- M Mathematics Is the solution shown below correct? Explain....
- E English What did the poem wistful (about the lonely hill) really mean?...
- B Biology (( NOT A QUESTION )) If you are having a bad day just know that I will sit here and let you vent your problems in my comment so if you need a place to just let it out, my comments...
- H History In this activity, you will draw maps that show the spread of Christianity in the ancient world. You may need to research and combine many different maps to get a full picture...

Ответ:
Approximately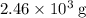 .
.
Explanation:
Molar mass of C₈H₁₈ and O₂Look up the following relative atomic mass values on a modern periodic table:
Calculate the molar mass of octane,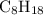 :
:
Similarly, calculate the molar mass of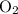 :
:
 .
.
Number of moles of octane moleculesHow many moles of molecules in of octane,
of octane, 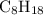 ?
?
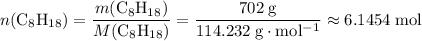 .
.
Number of moles of O₂ requiredMake sure that the chemical equation here is balanced.
In this equation,
The coefficient ofRatio between the two coefficients:
Therefore:
Rearrange to obtain:
It is already determined that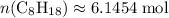 . Hence, the number of moles of oxygen gas required will be:
. Hence, the number of moles of oxygen gas required will be:
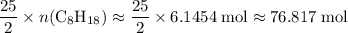 .
.
Mass of O₂ requiredWhat would be the mass of that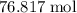 of
of 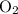 ?
?
Ответ:
B is 6.02x10^23
Explanation: