14 c > b + 14 n
6 7
what is the charge of a beta particle?
what is the mass of a beta particle?
Solved
Show answers
More tips
- C Computers and Internet How to insert videos into LiveJournal?...
- C Computers and Internet How Much Does an iPhone Cost in America?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- D Dating, Love, Relationships How Long Can Love Last?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- F Food and Cooking How to Find Your Zip Code?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
Answers on questions: Chemistry
- C Chemistry What is the mass of 0.25 mole of silvertrioxonitrate5...
- C Chemistry Your lab assistant rushes over to you. Terrified he tells you that the ideal gas you were monitoring has changed. Apparently the pressure had gone down (50 atm to 40 atm) but the...
- C Chemistry During electrolysis of molten Nacl.in presence of pt electrode the reaction takes place...
- C Chemistry Why can’t the calvin cycle run if the light reactions don’t occur?....
- C Chemistry If a water molecule h2o has two hydrogen atoms and one oxygen atom how would you describe the make up of a carbon dioxide molecule co2...
- C Chemistry Which wave would most likely be energetic enough to damage living human cells...
- C Chemistry If I have an ideal gas which is 2 liter in size and at 3 atmospheres of pressure. If the temperature changes from 50 to 250, and I also find that after this change it is 5 liters...
- C Chemistry As the atomic number increases in group 1, the number of valence electrons *blank* because...
- C Chemistry i need a answer to this. How many pieces of gum would you have to chew in order to ingest 1.00 mole of sucrose?...
- C Chemistry 2-propanol to 2-propanone ...

Ответ:
The number of moles of strontium bicarbonate is
Explanation:
Formula units are defined as lowest whole number ratio of ions in an ionic compound. It is calculate by multiplying the number of moles by Avogadro's number which is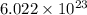
We are given:
Number of formula units of
As,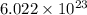 number of formula units are contained in 1 mole of a substance.
number of formula units are contained in 1 mole of a substance.
So, number of formula units will be contained in =
number of formula units will be contained in =  of strontium bicarbonate.
of strontium bicarbonate.
Hence, the number of moles of strontium bicarbonate is