tddreviews
20.01.2022 •
Chemistry
(2 pts) Determine the mass (in grams) of 9.33 L sample of xenon gas at STP
conditions. *
Your answer
This is a required question
Solved
Show answers
More tips
- A Art and Culture How to Learn to Sing? A Complete Guide for Beginners...
- H Health and Medicine How to Get Rid of Dandruff?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
- P Philosophy How did the concept of module arise in computer science?...
Answers on questions: Chemistry
- C Chemistry Which process takes a much longer time to produce organism with desirable trait as not all organism that are produced have desired genetic information...
- C Chemistry Anhydrous calcium chloride removes water vapor from the air inside a container having an air-tight seal called a desiccator. explain...
- C Chemistry Who wanna talk to me or play truth or dare...
- H History Was a leader in reforming the catholic church.....
- H History How did odysseus think the other beggar thought of him?...
- H History When the british declared martial law in massachusetts?...
- S Spanish How many tortillas does the average person in mexico consume? ?...
- S Social Studies What executive department is headed by the attorney general?...
- E English In the short story everybody use by alice walker what does the name dean represent to the narrator...
- H History In what ways does the united states constitution manifest the principles of both republican and democratic forms of government? in what ways does it deviate from...

Ответ:
The value of for the reaction will be -55.48 kJ
for the reaction will be -55.48 kJ
Explanation:
To calculate the number of moles, we use the equation:
Given mass of copper (I) oxide = 54.08 g
Molar mass of copper (I) oxide = 143.1 g/mol
Putting values in above equation, we get:
For the given chemical reaction:
By Stoichiometry of the reaction:
When 2 moles of copper (I) oxide reacts, the energy evolved is 292.0 kJ
So, when 0.380 moles of copper (I) oxide will react, the energy evolved will be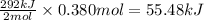
Sign convention of heat:
When heat is absorbed, the sign of heat is taken to be positive and when heat is released, the sign of heat is taken to be negative.
Hence, the value of for the reaction will be -55.48 kJ
for the reaction will be -55.48 kJ