iceecole6570
14.07.2020 •
Chemistry
250 g H 2 SO 4 completely reacted with aluminum?
2AI(s) + 32504(aq)- AI2 (504) 3(aq) + 3H 2 (9)
0.85g
450g
290g
870g
Solved
Show answers
More tips
- A Animals and plants 10 Tips for Growing Delicious and High-Quality Tomatoes in Your Garden...
- S Science and Technology Colliders: How They Work and Why They Matter...
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- W Work and Career Everything You Need to Know About MBA Programs...
- B Business and Finance What is the Difference Between Visa and Visa Gold?...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- S Sport When and Where Will the 2014 World Cup be Held?...
- S Sport When is the Champions League final?...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
Answers on questions: Chemistry
- C Chemistry What type of reaction is Cl2 + 2KBr = Br2 + 2KCl?...
- C Chemistry What is the chemical name of the compound cu2co3?...
- C Chemistry Determinar el volumen de 20 kg de benceno si su densidad es de 0,88g/cm3...
- C Chemistry Consider the half reactions below for a chemical reaction. Upper Z n right arrow upper Z n superscript 2 plus (a q) plus 2 e superscript minus. Second line: upper...
- E English What is a good description for Minho in The Maze Runner?...
- M Mathematics At the beginning of the year, Serenity had $90 in savings and saved an additional $13 each week thereafter. Andrew started the year with $40 and saved $18 every week....
- M Mathematics Help me asap, thank you!...
- M Mathematics I need help please it would mean alot...
- M Mathematics Find the slope of the line on the graph....
- H History Chat 4 me and richie plz dont report:(...

Ответ:
290 g of aluminium sulphate is produced.
Explanation:
To calculate the moles :
The balanced chemical reaction is:
According to stoichiometry :
3 moles of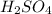 produce = 1 mole of
produce = 1 mole of 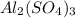
Thus 2.55 moles of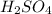 will require=
will require=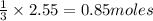 of
of 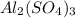
Mass of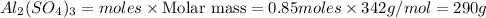
Thus 290 g of aluminium sulphate is produced.
Ответ:
Ca scans scan sk wo wka d Turkey show alone kar s
Explanation:
HHhah hahah ahhahh she sha aja aw