gjvyverman
30.03.2020 •
Chemistry
3. Determine if the overall solution containing the following salts would be acidic, basic or neutral. Write out the ion with their conjugates and state if the conjugate would be considered strong or weak. a. FeCl2 b. KHCO3 c. NH4F
Solved
Show answers
More tips
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- F Family and Home What is Most Important in Men s Lives?...
- G Goods and services Which TV is better - LCD or Plasma?...
- C Computers and Internet Are there special gaming mice?...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
- C Computers and Internet 3D Glasses! What is this thing?...
Answers on questions: Chemistry
- C Chemistry What is the volume in liters, of 0.50 mol of nitrogen gas(N2) at 25 C and 2.0 ATM?...
- C Chemistry A gas with a mass of 0.440 g has a volume of 0.224 L at STP what is the molar mass of the gas?...
- C Chemistry The equilibrium constant for the reaction shown below is Kc=0.0091. If the equilibrium concentrations of A and B are 0.031 M and 0.0823 M, respectively, what is the equilibrium...
- C Chemistry Using the formula n=c/v and the values from table 1, which material has the highest density?...
- C Chemistry What is the relative rate of effusion of Neon to carbon dioxide?...
- C Chemistry Zach invited his friend Sydney to a pool party over the weekend. Sydney informed Zach that the climate was supposed to be rainy over the weekend and suggested that he change...
- C Chemistry What is the charge on the nitride ion?...
- C Chemistry 2mg(s) + o2(g) → 2 mgo(s) δh = -1204 kj determine which statement is true regarding the reaction above. a) the reaction is endothermic; the products have a lower total bond...
- C Chemistry What is the total number of sublevels in the fourth principal energy level...
- C Chemistry What is the oxidation state for the first carbon in ch3cooh?...

Ответ:
The following solutions would be produced from dissociation of these salts: A. acidic B. Basic C. Acidic
Explanation:
Salts of weak bases and strong acids neutralization would hydrolyze, which gives it a pH less than 7. Salts formed by neutralization reaction between strong bases and weak acids hydrolyze, which gives it a pH greater than 7.A.) FeCl2 (aq) + H20 ⇄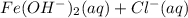 ------- weakly acidic
------- weakly acidic
B.) KHCO3(aq) + H20(aq) ⇄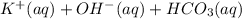 ----- strongly basic
----- strongly basic
C.) NH4F + H2O ⇄ -----weakly acidic
-----weakly acidic
This is due to the fact that the anion will become a spectator ion and fail to attract the H+, while the cation from the weak base will donate a proton to the water forming a hydronium ion.
Ответ:
C₂H₆ + 2Cl₂ => C₂H₄Cl₂ + 2HCl
Or from left to right:
1, 2, 1, 2
Explanation:
To balance an equation, start by counting the number of atoms of each element on both sides.
C₂H₆ + Cl₂ => C₂H₄Cl₂ + HCl
C 2 C 2
H 6 H 5
Cl 2 Cl 3
We should increase the number of hydrogen atoms (H) on the right (products).
C₂H₆ + Cl₂ => C₂H₄Cl₂ + 2HCl
C 2 C 2
H 6 H 6
Cl 2 Cl 4
Increase the number of chlorine atoms (Cl) on the left (reactants).
C₂H₆ + 2Cl₂ => C₂H₄Cl₂ + 2HCl
C 2 C 2
H 6 H 6
Cl 4 Cl 4
The equation is now balanced because there are the same number of atoms on each side.
C₂H₆ + 2Cl₂ => C₂H₄Cl₂ + 2HCl
or, if your drop down menus need you to write "1".
1C₂H₆ + 2Cl₂ => 1C₂H₄Cl₂ + 2HCl
The correct format is not to write the "1".