dakotacsey03
30.03.2020 •
Chemistry
3. Sulfite is a compound frequently added to preserve food and wine. In addition to inhibiting bacteria, it also prevents the oxidation of ethanol into acetic acid! a. It was previously discussed that sulfate, SO4 2– , can be categorized as a negligible base. Sulfite, SO3 2– , however, is basic. Considering the two structures, why is sulfite significantly more basic? b. Write the chemical equation and the equilibrium constant expression for the reaction between SO3 2– and water. c. Sulfurous acid, H2SO3 has a Ka1 = 1.23 x 10–2 and a Ka2 = 6.60 x 10–8 at 25 ºC. What is the pH of a 0.50M solution of Na2SO3?
Solved
Show answers
More tips
- T Travel and tourism Lost in the Catacombs: What to Do?...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
Answers on questions: Chemistry
- C Chemistry What is the total volume that you have if you calculated that it weighs 180g?...
- C Chemistry Х Densities of some of the substances are given in the table. Answer the following questions on the basis of them.If equal masses of all are taken, which one will have the largest...
- C Chemistry Which two gas samples have the same total number of molecules...
- C Chemistry The senior class at marion high school is ordering shirts...
- C Chemistry Which element, when combined with flourine, would most likely form an ionic compound? a.) lithium b.) carbon c.) phosphorus d.) chlorine...
- C Chemistry Why is the octet rule important in chemistry?...
- C Chemistry Which groups of metals tend to have more than one oxidation number?...
- C Chemistry If 5 mol of oxygen gas effuses through an opening in 10 seconds, how long will it take for the same amount of hydrogen gas to effuse under the same conditions?...
- C Chemistry What is covalent bond...
- M Mathematics For a random sample of 20 professional athletes, there is a strong, linear relationship between the number of hours they exercise per week and their resting heart rate. For the athletes...

Ответ:
a) The detailed explanation for the question being asked in section a is shown below
b)
Equilibrium constant expression![K_c = \frac{[HSO_3^-][OH^-]}{]SO_3^{2-}]}](/tpl/images/0571/3193/afa55.png)
c) pH = 10.45
Explanation:
a.
If we look at , we will realize that it is a conjugate base of a strong acid
, we will realize that it is a conjugate base of a strong acid 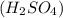 . However, the more stronger the acid, the weaker its conjugate base.
. However, the more stronger the acid, the weaker its conjugate base.
On the other hand is a conjugate base of a weak acid
is a conjugate base of a weak acid 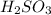 and the more weaker the acid, the stronger the basicity of its conjugate base.
and the more weaker the acid, the stronger the basicity of its conjugate base.
b. The chemical equation for the reaction between and water can be expressed as follows:
and water can be expressed as follows:
Equilibrium constant![K_c = \frac{[HSO_3^-][OH^-]}{]SO_3^{2-}]}](/tpl/images/0571/3193/afa55.png)
c.
The ICE Table is then constructed as follows:
Initial (M) 0.500 0 0
Change (M) - x + x + x
Equilibrium (M) (0.500 - x) x x
where is the ionic product of water =
is the ionic product of water = 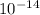
However;
pH + pOH = 14
pH + 3.55 = 14
pH = 14 - 3.55
pH = 10.45
Ответ:
50.0g C6H12O6 in 250g