nuggetslices
09.07.2019 •
Chemistry
352 ml of chlorine under a pressure of 720. mm hg are placed in a container under a pressure of 1120 mm hg. the temperature remains constant at 296 k. what is the volume of the container in liters? (0.226 l)
Solved
Show answers
More tips
- H Health and Medicine How to Get Rid of Dandruff?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
- P Philosophy How did the concept of module arise in computer science?...
Answers on questions: Chemistry
- C Chemistry When the universal indicator was added to the water was this a physical or chemical change...
- C Chemistry Which of these processes are chemical reactions? 1. boiling an egg 2. crushing spices 3. vegetables rotting 4. chopping vegetables 5. dew forming...
- C Chemistry Which statements describe the planets?...
- C Chemistry Find the molarity of a solution made by dissolving 17.6g of ZnCO3 with enough water to make 0.7500 Liters of soltuion....
- C Chemistry Which element below is Shiny, good conductor of heat and electricity, malleable and ductile? * 1 point Chlorine Nitrogen Magnesium Neon...
- C Chemistry If the theoretical yield of a reaction is 100 grams, which value for actual yield is physically impossible? a. 50 grams b. 99.9 grams c. 0 gramsd. 110 grams...
- C Chemistry • Explain, in terms of energy, particles motion & bonds, what happens when water is put into a freezer...
- C Chemistry How far does light travel in 2.5 seconds...
- C Chemistry How might a dependence on fossil fuels lead to international security risks? a. the greater demand for fossil fuels with decreasing supplies could lead to an increase in competitive...
- M Mathematics Ok but... what s 6+6? I ve been stuck on this forever and i can t see to find the answer. 0-0...

Ответ:
- 0.226 L
Solution:- From Boyle's law,"At constant temperature, volume of a gas is inversely proportional to the pressure of the gas."
Given:-
the equation could be rearranged for final volume as:
let's plug in the values in the equation and solve it for final volume.
Since, 1000 mL = 1 L
So,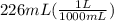
= 0.226 L
Hence, the volume of the container is 0.226 L.
Ответ:
ok so all you will have to do is multiplex
Explanation: