jjjjjjjbvhnchjkj
22.04.2020 •
Chemistry
4. When 1.00 L of 1.00 M Ba(NO3)2 solution at 25.0˚C is mixed with 1.00 L of 1.00 M Na2SO4 solution at 25.0˚C in a calorimeter, the white solid BaSO4 forms, and the temperature of the mixture increases. What is the final temperature of the mixture if the enthalpy change is – 26 kJ per mol BaSO4 formed? The specific heat of water is 4.18 J/g ·˚C, and the density of water is 1.00 g/mL
Solved
Show answers
More tips
- H Health and Medicine How to Increase Blood Pressure without Medication?...
- F Food and Cooking What to Give as a Wedding Gift?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- C Chemistry What is the valency of argon?...
- C Chemistry Please help fast What statement is correct about wind turbines. A. They create Mechanical and electrical energy B. They transform wind energy to electrical energy C. Their total...
- C Chemistry Use the reaction l2(s) = 12(9), AH = 62.4 kJ/mol, AS = 0.145 kJ/(mol-K), for this question At what temperature is the reaction at equilibrium? A. 157 K B. 62 K C. 0.002 K D....
- C Chemistry Which best describes the act of using senses or tools to gather information? creating a hypothesis making an observation summarizing the results recording the measurements...
- H History What claim can be made about the changes to the status of early Africans who arrived in Virginia?...
- M Mathematics 1991.475 to standard form...
- E English Write a essay about first day at school...
- C Chemistry Which method would you use to separate some salt from salty water...
- E English Refer to the Newsela article Climate Change in Hawaii and U.S. Tropical Islands. What is the author s purpose in mentioning wildlife in the article? A. to suggest that climate...
- H History Interpret religion in your own words....

Ответ:
The final temperature of the mixture is 28.11 °C
Explanation:
Step 1: Data given
Volume of 1.00 M Ba(NO3)2 = 1.00 L
Temperature = 25.0 °C
Volume of 1.00 M Na2SO4 = 1.00 L
enthalpy change is – 26 kJ per mol BaSO4
The specific heat of water is 4.18 J/g ·˚C
the density of water is 1.00 g/mL
Step 2: The balanced equation
Ba(NO3)2(aq) + Na2SO4(aq) → 2NaNO3(aq) + BaSO4(s)
Step 3: Calculate the total volume
Total volume = 1.00 L + 1.00 L = 2.00 L = 2000 mL
Step 4: Calculate mass
Mass = volume * density
Mass = 2000 mL * 1g/mL
Mass = 2000 grams
Step 5: Calculate moles BaSO4 formed
For 1 mol Ba(NO3)2 we need 1 mol Na2SO4 to produce 1 mol BaSO4
There is no limiting reactant, both Ba(NO3)2 and Na2SO4 will be completely be consumed (1 mol). We'll have 1.0 mol of BaSO4 produced.
Step 6: Calculate Q
Q = - ΔH
ΔH is negative so the reaction is exothermic, what means the temperature increases
Q is always positive, so Q = 26kJ = 26000 J
Step 6: Calculate the heat transfer
Q= m*c*ΔT
⇒with Q = the heat transfer = TO BE DETERMINED
⇒with m =the mass of the solution = 2000 grams
⇒with c= the specific heat of the solution = 4.18 J/g°C
⇒with ΔT = the change of temperature = T2 - T1 = T2 - 25.0
26000 = 2000 * 4.18 * (T2 - 25.0 °C)
3.11 = T2 - 25.0 °C
T2 = 25.0 + 3.11 °C
T2 = 28.11 °C
The final temperature of the mixture is 28.11 °C
Ответ:
Explanation:
From the information given:
Mass of carbon tetrachloride = 5 kg
Pressure = 1 bar
The given density for carbon tetrachloride = 1590 kg/m³
The specific heat of carbon tetrachloride = 0.84 kJ/kg K
From the composition, the initial volume of carbon tetrachloride will be: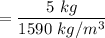
= 0.0031 m³
Suppose is independent of temperature while pressure is constant;
is independent of temperature while pressure is constant;
Then:
The change in volume can be expressed as:
However; the workdone = -PdV
W = - 7.6 J
The heat energy Q = Δ h
Q = 84 kJ
The internal energy is calculated by using the 1st law of thermodynamics; which can be expressed as;
ΔU = ΔQ + W
ΔU = 84 kJ + ( -7.6 × 10⁻³ kJ)
ΔU = 83.992 kJ