naiomireyes74p2aybs
26.03.2020 •
Chemistry
5. Azulene is a beautiful blue hydrocarbon. If 0.106 g of the compound is burned in oxygen, 0.364 g of CO2 and 0.0596 g of H2O are isolated. Determine its empirical and molecular formulas if the molar mass of azulene was found to be 128.2 g. Is it an alkane
Solved
Show answers
More tips
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
- F Food and Cooking Homemade French Fries: The Ultimate Guide...
- D Dating, Love, Relationships How Long Can Love Last?...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- A Auto and Moto Discovering the Leader: What is the Most Expensive Car in the World?...
- F Food and Cooking How to Quickly Put your Child to Sleep?...
- C Computers and Internet How to Create a Website for Free and Easy?...
Answers on questions: Chemistry
- E English What is the theme of the story beautiful girl but Tobias Wolff?...
- E Engineering A Carnot refrigerator absorbs heat from a space at 15°C at a rate of 16,000 kJ/h and rejects heat to a reservoir at 36°C. page 315Determine the COP of the refrigerator,...
- M Mathematics Gail has 2 gallons of milk. About how many liters of milk does Gail have?(1 quart =0.94 liters)...
- M Mathematics Find the area of triangle ABC and the area of XYZ. AABC - AXYZ....

Ответ:
The empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.
Explanation:
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, 'x' and 'y' are the subscripts of Carbon, hydrogen and oxygen respectively.
We are given:
Mass of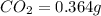
Mass of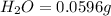
We know that:
Molar mass of carbon dioxide = 44 g/mol
Molar mass of water = 18 g/mol
For calculating the mass of carbon:
In 44 g of carbon dioxide, 12 g of carbon is contained.
So, in 0.364 g of carbon dioxide, of carbon will be contained.
of carbon will be contained.
For calculating the mass of hydrogen:
In 18 g of water, 2 g of hydrogen is contained.
So, in 0.0596 g of water,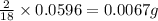 of hydrogen will be contained.
of hydrogen will be contained.
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.Moles of Carbon =
Moles of Hydrogen =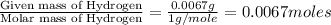 ]
]
Step 2: Calculating the mole ratio of the given elements.For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0067 moles.
For Carbon =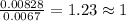
For Hydrogen =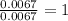
Step 3: Taking the mole ratio as their subscripts.The ratio of C : H = 1 : 1
The empirical formula for the given compound is CH
For determining the molecular formula, we need to determine the valency which is multiplied by each element to get the molecular formula.
The equation used to calculate the valency is:
We are given:
Mass of molecular formula = 128.2 g/mol
Mass of empirical formula = 13 g/mol
Putting values in above equation, we get:
Multiplying this valency by the subscript of every element of empirical formula, we get:
The general formula of an alkane is , where n = any natural number
, where n = any natural number
Here, n = 10 and it does not satisfy being an alkane
Hence, the empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.
Ответ:
he probably just wanted to toggling with u or prob he just wanted u to fall for him and then leave. But prob he got a girl already, hope this helps :D
Explanation: