5. What is the mathematical equation used when solving calorimetry problems? What does
each variable in the equation represent?
Solved
Show answers
More tips
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- M Mathematics Find the area of the region bounded by the graphs of y = x, y = 4 − 3x, and x = 0. 0.375 2 4 none of these...
- M Mathematics A restaurant wants to study how well its salads sell. The circle graph shows the sales over the past few days. If 40 of the salads sold were Caesar salads how many total...
- B Business Pizza hut provides the use of its name plus operating know-how to companies in costa rica in return for a fee plus a percentage of profits. pizza hut is...

Ответ:
a)
The overall balanced combustion reaction is written as :
b)
the higher heating values per unit mass of LPG = 49.9876 MJ/kg
per unit mass of LPG = 49.9876 MJ/kg
the lower heating values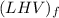 per unit mass of LPG = 46.4933 MJ/kg
per unit mass of LPG = 46.4933 MJ/kg
Explanation:
a)
The stoichiometric equation can be expressed as :
Now, equating the coefficient of carbon; we have:
(0.7×3)+(0.05×4)+(0.25×3) = a
a = 3.05
Also, Equating the coefficient of hydrogen : we have:
(0.7 × 8) +(0.05 × 10) + ( 0.25 × 6) = 2 b
2b = 7.6
b = 3.8
Equating the coefficient of oxygen
2x = 2a + b
Equating the coefficient of Nitrogen
Therefore, The overall balanced combustion reaction can now be written as :
Now; To determine the stoichiometric F/A and A/F ratios; we have:
b)
What are the higher and lower heating values per unit mass of LPG?
Let calculate the molecular mass of the fuel in order to determine their mass fraction of the fuel components.
Molecular mass of the fuel
= 30.8 + 2.9 + 10.5
= 44.2 kg/mol
Mass fraction of the fuel components can now be calculated as :
Finally; calculating the higher heating values per unit mass of LPG; we have:
per unit mass of LPG; we have:
calculating the lower heating values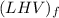 per unit mass of LPG; we have:
per unit mass of LPG; we have: