luccasmith6764
08.07.2019 •
Chemistry
6. how to calculate the mass of copper that can be plated out of a 1.0 m cu(no3)2 solution using a current of 0.75 a for 5.0 minutes?
Solved
Show answers
More tips
- H Health and Medicine What You Need to Know About Nasal Congestion in Infants: Causes, Symptoms, and Treatment...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- C Computers and Internet How to Choose an Uninterruptible Power Supply (UPS) for Your Computer: Expert Tips...
- S Science and Technology How to choose a home theater system?...
- A Auto and Moto How to Choose a Car Wash? Tips and Recommendations...
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- S Sport When is the Champions League final?...
Answers on questions: Chemistry
- C Chemistry How much heat is absorbed by 20g granite boulder as energy from the sun causes its temperature to change from 10°C to 29°C? (Specific heat capacity of granite is 0.1 cal/goC)...
- C Chemistry When a mixture of 12.0 g of acetylene (c2h2 and 12.0 g of oxygen (o2 is ignited, the resultant combustion reaction produces co2 and h2o?...
- E English What is the subject and authors attitude towards subject for the following selections; mrs. flowers, ishi two worlds, and Harriet Tubman Describe the setting of the following...
- M Mathematics Please please help me...
- B Business The following events apply to Lewis and Harper, a public accounting firm, for the Year 1 accounting period: Performed $70,000 of services for clients on account. Performed...
- P Physics Precisely 1.00 s after the speeder passes, the police officer steps on the accelerator; if the police car s acceleration is 2.70 m/s2 , how much time passes after the police...
- L Law Former Major League Baseball player and radio game announcer Bob Uecker sought an injunction against Ann Ladd, alleging a six- or seven-year pattern of harassment. Ladd, who...
- M Mathematics Mr.Cantu is selling snow cones. He used 216.2 ounces of ice to make 47 snow cones. If he used an equal amount of ice in each one, how many ounces of ice are in each one...
- B Biology So nobody gonna help me Okay...
- H History HELP ASAP WORTH 10 POINTS FIRST PERSON TO ANSWER CORRECTLY GETS BRAINLIEST Which reasons explain the failure of the League of Nations? Select two options. A. The League of...

Ответ:
Approximately 0.074 grams.
Explanation:
Look up the relative atomic mass of copper on a modern periodic table:How does the electroplating works for copper? Copper exists as copper(II) ions in the copper(II) nitrate
in the copper(II) nitrate 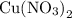 solution. It takes two moles of electrons to reduce one mole of copper(II) ions
solution. It takes two moles of electrons to reduce one mole of copper(II) ions  to metallic copper
to metallic copper 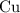 .
.
 . (Reduction half of the ionic equation.)
. (Reduction half of the ionic equation.)
What are the steps for finding the mass of copper that has been deposited.Start by finding the charge on the electrons that have been supplied to this electrochemical cell. After that,
Find the number of moles of electrons that have been supplied based on the charge supplied; Find the number of moles of copper that have been reduced based on the number of moles of electrons supplied; andFind the mass of copper based on the number of moles of copper atoms reduced.What's the chargeFor a constant direct current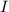 , the charge
, the charge 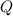 that has been delivered in time
that has been delivered in time  is equal to
is equal to
In case
For this electrochemical cell,
 .
.
How many moles of electrons were supplied to this electrochemical cell?The Faraday's Constant gives the size of charge (in Coulombs) on one mole of electrons.
 .
.
How many moles of copper atoms have been deposited?Assume that copper(II) ions are in excess. Refer to the reduction half-equation, it takes two moles of electrons to deposit one mole of metallic copper.
are in excess. Refer to the reduction half-equation, it takes two moles of electrons to deposit one mole of metallic copper.
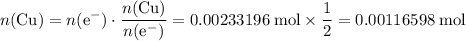 .
.
What's the mass of that many copper atoms?The Relative atomic mass of copper is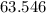 .
.
Ответ: