8.
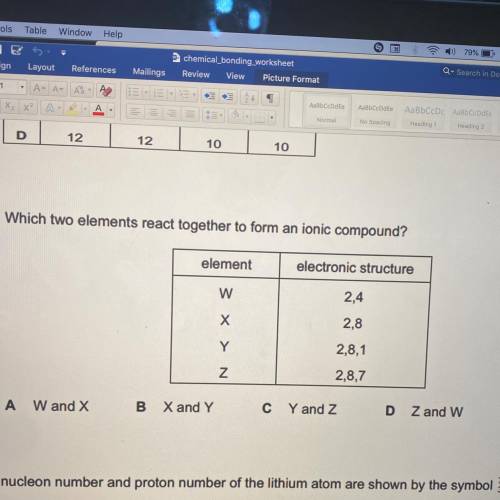
Which two elements react together to form an ionic compound?
element
electronic structure
2,4
11 pm
2,8
3 X > N
2,8,1
2,8,7
A
W and X
B
X and Y
C Y and Z
D
Z and w
Solved
Show answers
More tips
- F Food and Cooking How to Make Delicious Plov: Secrets and Recipes...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- C Chemistry What happens when light strikes a dark heavy object...
- C Chemistry A10.0-gram sample of h2o(l) at 23.0°c absorbs 209 joules of heat. what is the final temperature of the h2o(l) sample? (1) 5.0°c (3) 28.0°c(2) 18.0°c (4) 50.0°c...
- C Chemistry Which one of the following is the symbol used in a chemical equation for a gas? A. (g) B. (aq) C. (gas) D. (a)...
- C Chemistry A group of kids trick or treating are dressed as a bunny rabbit, sunflower and a wolf. Which of these costumes represents a primary producer? E. sun F. sunflower G rabbit H. Wolf...
- C Chemistry Can sum1 Help me with my CHEMISTRY ANYONE PLEASE...
- C Chemistry Both pyrrole and pyridine are aromatic compounds, and undergo electrophilic aromatic substitution (EAS). Using a resonance argument, predict the regiochemistry of EAS on both pyrrole...
- C Chemistry An example of a substance that is a strong base would be . Bleach Water Milk Vinegar...
- C Chemistry 4. An 82 kg hiker climbs Mt. Humphrey near Flagstaff. During a two hour period, the hiker s vertical elevation increases by 540 meters, a. Calculate the climber s delta Eg...
- C Chemistry Please help- science 1. - new 2. full 3. first quarter 4. last quarter...
- C Chemistry Click an item in the list or group of pictures at the bottom of the problem and, holding the button down, drag it into the correct position in the answer box. Release your mouse...

Ответ:
A. 0.0625 moldm¯³
B. 6.125 gdm¯³
Explanation:
We'll begin by writing the balanced equation balance equation for the reaction. This is illustrated below:
2NaOH + H₂SO₄ —> Na₂SO₄ + 2H₂O
From the balanced equation above,
The mole ratio of the acid, H₂SO₄ (nₐ) = 1
The mole ratio of the base, NaOH (n₆) = 2
A. Determination of the concentration of the acid in moldm¯³.
Volume of base, NaOH (V₆) = 20 cm³
Concentration of base, NaOH (C₆) = 0.2 moldm¯³
Volume of acid, H₂SO₄ (Vₐ) = 32 cm³
The mole ratio of the acid, H₂SO₄ (nₐ) = 1
The mole ratio of the base, NaOH (n₆) = 2
Concentration of acid, H₂SO₄ (Cₐ) =?
CₐVₐ / C₆V₆ = nₐ / n₆
Cₐ × 32 / 0.2 × 20 = 1 / 2
Cₐ × 32 / 4 = 1 / 2
Cross multiply
Cₐ × 32 × 2 = 4 × 1
Cₐ × 64 = 4
Divide both side by 64
Cₐ = 4 / 64
Cₐ = 0.0625 moldm¯³
Therefore, concentration of the acid in moldm¯³ is 0.0625 moldm¯³
B. Determination of the concentration of the acid in gdm¯³.
Concentration (moldm¯³) = 0.0625 moldm¯³
Concentration (gdm¯³) =?
Next, we shall determine the molar mass of the acid, H₂SO₄. This can be obtained as follow:
Molar mass of H₂SO₄ = (2×1) + 32 + (4×16)
= 2 + 32 + 98
= 98 g/mol
Finally, we shall determine the concentration of the acid in gdm¯³. This can be obtained as follow:
Concentration (moldm¯³) = 0.0625 moldm¯³
Molar mass of H₂SO₄ = 98 g/mol
Concentration (gdm¯³) =?
Conc. (moldm¯³) = conc. (gdm¯³) / Molar mass
0.0625 = Conc. (gdm¯³) / 98
Cross multiply
Conc. (gdm¯³) = 0.0625 × 98
Conc. (gdm¯³) = 6.125 gdm¯³
Thus, the concentration of the acid in gdm¯³ is 6.125 gdm¯³