A 216 mL sample of gas is at STP. What volume will it occupy at 975 torr?
Solved
Show answers
More tips
- L Leisure and Entertainment Unlocking the Secrets of Fast and Effective Tectonic Learning...
- S Style and Beauty How to Choose the Perfect Hair Color?...
- C Computers and Internet Best iPad Games: Our Opinion...
- A Animals and plants Man s Best Friend: Which Dog Breed Is the Most Friendly?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry Which type of luster gives the mineral the appearance of metal?...
- C Chemistry Calculate ΔS∘rxn for the balanced chemical equation 2H2S(g)+3O2(g)→2H2O(g)+2SO2(g) Express the entropy change to four significant figures and include the appropriate...
- C Chemistry Sodium forms a cation with net charge of +1. How many sodium ions can bond with carbonate? answers a.2 b.1 c.4 d.3...
- C Chemistry Which of the following elements can t have an expanded octet? answers A. oxygen B. phosphorous C. chlorine d. sulfer...
- C Chemistry Using the standard entropy values: C2H4(g), S° = +219.8 J mol-1 K-1 H2(g), S° = +130.6 J mol-1 K-1 C2H6(g), S° = +229.5 J mol-1 K-1 calculate the standard entropy change,...
- C Chemistry Of the following, which are accurate statements regarding buffer capacity? Select all that apply: If enough base is added to a buffer to react with all of the weak acid...
- C Chemistry What is five plus forty five...
- C Chemistry HELP What happens when nitrogen fills its valence shell?Three electrons are lost, creating N+3.Three electrons are gained, creating N−3.Three electrons are lost, creating...
- C Chemistry Mira is testing objects for her science experiment. She has a chart listing the mystery objects densities, but no other information. Object Density A 12 g/cm³ B 9 g/cm³...
- C Chemistry What is the mass in grams of 31.0 mL of gasoline?...

Ответ:
C) hydrochloric acid and, in case that the question allows for more than one choices, D)
hydrochloric acid and, in case that the question allows for more than one choices, D)  carbon dioxide as well.
carbon dioxide as well.
Methane molecules are nonpolar and barely dissolve in water.
are nonpolar and barely dissolve in water.
Sodium hydrocarbonate undergoes hydrolysis to release hydroxide ions
undergoes hydrolysis to release hydroxide ions  that can end up consuming
that can end up consuming 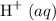 :
:
Hydrochloric acid, a strong acid, ionizes to produce protons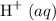 and chloride ions when dissolved in water:
and chloride ions when dissolved in water:
Carbon dioxide reacts with water to produce hydrocarbonic acid, a weak acid that slightly ionizes, also producing protons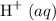 but to a significantly lesser extent than hydrochloric acid does.
but to a significantly lesser extent than hydrochloric acid does.