nanamath5662
07.03.2020 •
Chemistry
A chemist dissolves of pure nitric acid in enough water to make up of solution. Calculate the pH of the solution. Be sure your answer has the correct number of significant digits.
Solved
Show answers
More tips
- S Society and Politics Why Did Medvedev Announce a New Privatization of State Property?...
- F Family and Home Stay Warm but Don t Overheat: What is the Optimal Temperature for Your Home During Winter?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...

Ответ:
The given question is incomplete. The complete question is
A chemist dissolves 314 mg of pure nitric acid in enough water to make up 190 ml of solution. Calculate the pH of the solution. Be sure your answer has the correct number of significant digits.
Explanation:
Molarity of a solution is defined as the number of moles of solute dissolved per Liter of the solution.
where,
n= moles of solute=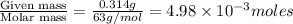
pH or pOH is the measure of acidity or alkalinity of a solution.
pH is calculated by taking negative logarithm of hydrogen ion concentration.
1 mole of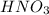 gives 1 mole of
gives 1 mole of 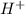
Thus 0.026 moles of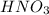 give=
give= 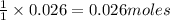 of
of 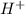
Putting in the values:
Thus pH of the solution is 1.58
Ответ:
hi
Explanation: