mbonham481
05.05.2020 •
Chemistry
A compound has the following percent composition: nitrogen (9.66%), hydrogen (2.79%) and iodine (87.6%). What is the empirical formula?
Solved
Show answers
More tips
- L Leisure and Entertainment How to Find a Phone Number by Address: The Ultimate Guide...
- P Philosophy How to Properly Create a Vision Board?...
- C Computers and Internet What is Web 2.0 and How Does it Work?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- C Chemistry Which statement best describes the relationship between the giraffe and the tree shown above? The giraffe survives by getting food and oxygen from the tree. * The giraffe survives...
- C Chemistry The weather characteristics of air mass results primarily form its. 1. geographic origin. 2. size and shape. 3. rate of movement. 4. direction of movement....
- C Chemistry Guess my name of u gras it u get 100 points...
- C Chemistry WHAT IS DIRECT SPEECH??plz URGENT.....
- C Chemistry What does AS 0 mean in a reaction? A. The reaction was exothermic. B. The reaction increased in order. C. The reaction is at equilibrium. D. The reaction was endothermic....
- C Chemistry Which of the following statements correctly describes the relationship between volume (v) and number of moles (n) illustrated in the following plot for a gas constant temperature...
- C Chemistry What happens when the atoms of a substance are regrouped?...
- C Chemistry When the ozone reacts with chlorine what is the resulting product formulas...
- C Chemistry Can someone help on this?...
- C Chemistry 3. If, in the reaction below, 49 g of Fe,produces a 78.25 % yield of Fe. How many grams are produced?Fe,O. + 4 H2 - 3 Fe + 4 H,0lilleEL...

Ответ:
Explanation:
The given data is as follows.
Density of vinegar = 1.0 g/ml
Specific heat capacity = 4.25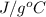
Relation between enthalpy and specific heat is as follows.
Hence, putting the values into the above formula as follows.
= (as density =
(as density = 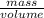 )
)
= - 315 J
Thus, we can conclude that the enthalpy of reaction is -315 J.
As the value is negative so, it means that heat is releasing. Hence, the reaction is exothermic in nature.