norahramahi123
03.12.2020 •
Chemistry
A cylinder, with a piston pressing down with a constant pressure, is filled with 2.10 moles of a gas (n1), and its volume is 46.0 L (V1). If 0.700 mole of gas leak out, and the pressure and temperature remain the same, what is the final volume of the gas inside the cylinder?
Solved
Show answers
More tips
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry How many moles of each molecule are represented in the equation, 2H2 + O2 + 2H20? A 2, 1, 1 B 2, 2, 2 C 2, 1, 2 D 1, 1, 1...
- M Mathematics What is the approximate volume of a cone with a height of 9 ft and radius of 3 ft? use 3.14 to approximate pi, and express your final answer to the nearest hundredth. i got the...
- M Mathematics The following diagram displays a disposable mold that a company uses to craft gold cones used in jewelry. Each mold is only used once. Molds are made by a 3D printer. What is...
- M Mathematics Last set of 100 points...

Ответ:
The volume is
Explanation:
From the question we are told that
The number of moles of the gas is
The volume of the gas is
The of moles of the gas that leaked is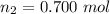
Generally from the ideal gas law
Generally at constant pressure and temperature
So
=>
=>
=>
Ответ:
Combination reaction