jabper5522
02.03.2020 •
Chemistry
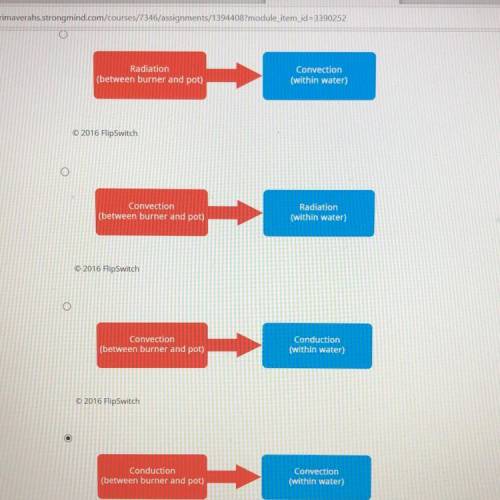
A metal pot of water is put on a stove. The stove heats up and the water boils. The system consists of the stove, water, and the pot. Which of the following flowcharts most accurately models the path of heat energy transferred in this system?
Solved
Show answers
More tips
- P Philosophy Is Everything We Strive for Eventually Achieved and Destroyed?...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
Answers on questions: Chemistry
- C Chemistry The placement of the double bond goes between which two atoms in this formaldehyde molecule? note: hydrogen, carbon, and oxygen have one, four, and six valence electrons...
- C Chemistry The ph of a solution is 4.36. what is the poh?...
- C Chemistry Use the activity series Au + Pd(NO3)2...
- C Chemistry At these conditions, the binary molecular diffusion coefficient of CO2 in air is 0.161 cm2 /s, but the effective diffusion coefficient of CO2 within the porous medium...
- C Chemistry The uneven heating of the atmosphere by the sun s energy creates pressure differences. Which effect is a direct result of this? formation of clouds O generation of winds...
- C Chemistry The dehydration butanol on alumina is carried out over a silica-alumina catalyst at 680 K. CH3CH2CH2OH →CH3CH = CHCH3 + H2O The rate law iswith k = 0.054 mol/gcat·h·atm...
- C Chemistry how the charge of an element is used to determine the chemical formula if the elements have equal and unequal charges....
- C Chemistry A 3.458 g sample of KHP, a monoprotic acid, requires 45.71 mL of a KOH solution to reach the endpoint. What is the concentration of the KOH solution...
- P Physics Acar is traveling on a freeway onramp at 25 mph. the pavement s coefficient of road adhesion is 0.80. the car has cd 0.40, a,-20.5 ft, and w 2200 lb. the wheelbase is...
- P Physics What is the instantaneous velocity of a freely falling object 9.0 s after it is released from a position of rest? express your answer to two significant figures and include...

Ответ:
Yeah, you're correct.
Explanation:
Convection when the water kind of "moves" within the pot and conduction when the pot is touching the stove.
Ответ:
Balanced equation: H₂S₂O₇ + H₂O → 2H₂SO₄
Theoretical yield of H₂SO₄: 1322 g
Percent yield for the H₂SO₄ production: 33%
Explanation:
First of all, we determine the balanced equation:
H₂S₂O₇ + H₂O → 2H₂SO₄
Stoichiometry says that 1 mol of oleum reacts to 1 mol of water in order to produce 2 moles of sulfuric acid.
1.20 kg = 1200 g
We convert the grams to moles → 1200 g . 1mol/ 178.12g = 6.74 mol
Ratio is 1:2, so if we have 6.74 mol of oleum we can produce the double of moles of acid. (6.74 . 2) = 13.48 moles of acid.
This is the 100 % yield reaction, or it can be called theoretical yield.
We convert the moles to mass → 13.48 mol . 98g /1mol = 1322 g of acid may be produced.
In the problem, they said we produced 435 g of acid, so let's determine the percent yield.
Percent yield of reaction = (Produced yield / Theoretical yield) . 100
Percent yield of reaction = (435 / 1322) . 100 → 33 %