KingMack1136
20.03.2020 •
Chemistry
A mixture consisting of 0.250 M N2(g) and 0.500 M H2(g) reaches equilibrium according to the equation N2(g) + 3 H2(g) → 2 NH3(g). At equilibrium, the concentration of ammonia is 0.150 M. Calculate the concentration of H2(g) at equilibrium.
Solved
Show answers
More tips
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
Answers on questions: Chemistry
- C Chemistry Is zn2+ a transition metal?...
- C Chemistry Who is obama? zzznnxnz...
- C Chemistry Δh o f of hydrogen chloride [hcl(g)] is −92.3 kj/mol. given the following data, determine the identity of the two missing products and calculate the δhrxn for equation 3....
- M Mathematics Which statement best describes the function below? f(x) = 2x^3 + 2x^2 –x O A. It is a one-to-one function. O B. It is a many-to-one function. O c. It is not a function. OD....
- M Mathematics Janet has a awake drawing of her room 3.4 in to 2.8 in if each inch om a scale drawing equalss 5 feet what are the actual dimensions of Janet s room?...
- M Mathematics If two lines intersect, then they what in exactly one point...
- M Mathematics If you get this right, you will be brainliest!! What would be the equivalent to y=13? (Disclaimer: This is literally it, no graph, no multiple-choice, no nothing. I m really...
- E English How am I not gay if my grandpa is?...
- S Social Studies These 2 questions are what I need help on...
- E English I need help please. Ill do anything if you guys please tell me...

Ответ:
The concentration of H2 at the equilibrium is 0.275 M
Explanation:
Step 1: Data given
Molarity of N2 = 0.250 M
Molarity of H2 = 0.500 M
At equilibrium, the concentration of ammonia is 0.150 M
Step 2: The balanced equation
N2(g) + 3 H2(g) → 2 NH3(g)
Step 3: The initial concentrations
[N2] = 0.250 M
[H2] = 0.500 M
[NH3] = 0 M
Step 4: The concentrations at the equilibrium
[N2] = 0.250 - X M
[H2] = 0.500 - 3X M
[NH3] = 2X M = 0.150
X = 0.150 / 2 = 0.075
[N2] = 0.250 - 0.075 M = 0.175 M
[H2] = 0.500 - 3X M = 0.275 M
[NH3] = 2X M = 0.150
Step 5: Calculate Kc
Kc = [NH3]² / [N2][H2]³
Kc = (0.150²) / (0.175 * 0.275³)
Kc = 1.70
The concentration of H2 at the equilibrium is 0.275 M
Ответ:
The mass of sample is 21.6 grams.
Explanation:
We are given:
Volume of cylinder without object,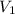 = 10 mL
= 10 mL
Volume of cylinder with object,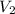 = 18 mL
= 18 mL
Volume of object =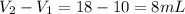
To calculate volume of a substance, we use the equation:
We are given:
Density of sample (aluminum) = 2.7 g/mL
Volume of sample (aluminum) = 8 mL
Putting values in above equation, we get:
Hence, the mass of sample is 21.6 grams.