lovelyheart5337
14.06.2021 •
Chemistry
A mixture of coarse sand and sugar is 45.0 percent sand by mass. 120.0 grams (g) of the mixture is placed in a fine-mesh cloth bag and dunked repeatedly in Lake Michigan. After drying, the mass of the contents of the bag equals: .
A. 66.0 g
B. 120.0 g
C. 65.0 g
D. 72.00 g
E. 54.0 g
Solved
Show answers
More tips
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- S Science and Technology Colliders: How They Work and Why They Matter...
- A Animals and plants Unraveling the Mystery of Loch Ness: What Does the Loch Ness Monster Look Like?...
- L Leisure and Entertainment How Many Seasons are There in the TV Show Interns?...
- S Sport Playing Bowling: Rules and Advice for Novices...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- C Computers and Internet What is Web 2.0 and How Does it Work?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- C Chemistry I need help I don’t understand it and I need it done before the end of today...
- C Chemistry Hydrogen and oxygen undergo a chemical reaction to form water. how much water would form if 2.56 g of hydrogen reacted completely with 20.32 g of oxygen? g...
- B Biology 1. Healthy water will have a (high / low) variety of bioindicators, while a polluted body of a water will have a (high / low) variety 2. What four things impact the amount...
- E English Please, Help me! Turn these requests, commands and instructions into reported speech. 1. ‘Please, please don’t call my mother!’ the boy said to them. — The boy begged them...

Ответ:
Option E
Explanation:
From the question we are told that:
Amount of sand in percentage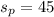
Sample size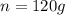
Note:After being dumped in the river repeatedly the sugar melts away leaving behind the insoluble sand
Generally the equation for Amount of sand content is mathematically given by
Therefore
After drying, the mass of the contents of the bag equals
Option E
Ответ:
Number of electrons in outer shell determines atom reactivity, not number of protons and neutrons.
Electron configuration of fluorine: ₉F 1s² 2s² 2p⁵.
Electron configuration of neon: ₁₀N 1s² 2s² 2p⁶.
Fluorine tends to have eight electrons in outer shell like neon (noble gas) and gains one electron in chemical reaction.