alejandra216
27.08.2020 •
Chemistry
A reaction between 7.0 g of copper(II) oxide and 50 mL of 0.20 M nitric acid produces
copper(II) nitrate, Cu(NO3)2 and water.
(c) Determine the limiting reactant.
(d) Calculate the mass of excess reactant after the reaction.
(ANS: 6.6068g)
(e) Determine the percentage yield if the actual mass of copper (II) nitrate obtained from
the reaction is 0.85 g.
(ANS: 90.64%)
How to get the mass of HNO3 from here? I only managed to get mass of NO3 based on the molarity formula. thanks!
Solved
Show answers
More tips
- S Science and Technology How to Secure Exam Sessions: Silence Mobile Phones in the Classroom...
- G Goods and services How to Choose a Bread Maker?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry When does ice start to melt...
- C Chemistry Here are the right answersDrag the item from the item bank to its corresponding match....
- C Chemistry Can relative mass help you count by weighing or get a relative count of things?...
- C Chemistry There is a high risk of earthquakes along the San Andreas fault in California. What is happening in Earth s crust along the fault to cause this high earthquake risk? Use the...
- C Chemistry Which planetary body has the greatest gravitational pull? a) jupiter b) neptune c) the moon d) the earth...
- C Chemistry Ahorizontal row of elements in the periodic table is known as a group or period...
- C Chemistry Calculate the density of sulfuric acid if 35.4 ml of the acid weighs 65.14 g....
- C Chemistry A 6.2-L balloon at 55˚C and 615mm Hg is taken to the top of Mt. Everest (-35˚C and 400- mmHg). What is the size of the balloon?...
- C Chemistry Scandium oxide is insoluble in water. Describe how you could show that it is an amphoteric oxide...
- C Chemistry Determine the maximum pressure of water vapor in wet hydrogen at 1 atm pressure in which chromium can be heated without oxidation occurring at 1500 K. Is the oxidation of...

Ответ:
(c) The limiting reactant is HNO₃
(d) The mass of the excess reactant after the reaction is approximately 6.6 grams
(e) The percentage yield of copper (II) nitrate from the reaction is approximately 90.64%
The reason the above values are the correct values is as follows;
The given parameters are;
The mass of copper(II)oxide in the reaction = 7.0 g
The volume of the 0.20 M nitric acid, HNO₃ = 50 mL
(c) Concentration of the reactants
The molar mass of CuO = 79.545 g/mol
Number of moles = Mass/(Molar mass)
The number of moles of CuO = (7 g)/79.545 g/mol ≈ 0.088 moles
50 mL of 0.20 M HNO₃, contains 50/1000 × 0.2 = 0.01 moles of HNO₃
The chemical equation for the reaction is CuO + 2HNO₃ → Cu(NO₃)₂ + H₂O
Therefore;
One mole of CuO reacts with two moles of HNO₃ to produce one mole of Cu(NO₃)₂ and one mole of H₂O
Therefore, 0.088 moles of CuO reacts with 2 × 0.088 = 0.176 moles of HNO₃
Given that there is only 0.01 moles of HNO₃, the limiting reactant is the HNO₃, which is not enough to completely react with the CuO which is the excess reactant
(d) The mass of the CuO that reacts with the 0.01 moles of HNO₃ is given as follows;
1 mole of CuO reacts with 2 moles HNO₃
0.01 moles of HNO₃ will react with 0.01/2 = 0.005 moles of CuO
Mass = Number of moles × Molar mass
The mass of 0.005 moles of CuO = 0.005 moles × 79.545 g/mol = 0.397725 grams
The mass of the CuO left = Initial mass - Reacting mass
∴ The mass of the CuO left = 7 grams - 0.397725 grams = 6.602275 grams
The mass of the excess reactant (CuO) after the reaction ≈ 6.6 grams
(e) The theoretical number of moles of copper (II) nitrate, Cu(NO₃)₂ produced = Half the number of moles of HNO₃ in the reaction
The number of moles of HNO₃ in the reaction = 0.01 moles
∴ The theoretical number of moles of copper (II) nitrate, Cu(NO₃)₂ produced = (1/2) × 0.01 moles = 0.005 moles
The molar mass of Cu(NO₃)₂ = 187.56 g/mol
The theoretical mass of Cu(NO₃)₂ produced = 0.005 moles × 187.56 g/mol = 0.9378 grams
The actual yield of copper (II) nitrate is 0.84 g
Therefore;
The percentage yield of copper (II) nitrate, %yield ≈ 90.64%
Learn more about percentage yield from chemical reactions here:
link
Ответ:
(c) Nitric acid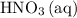 is the limiting reactant.
is the limiting reactant.
(d) Approximately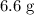 of
of 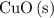 will be in excess.
will be in excess.
(e) The percentage yield of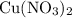 is approximately
is approximately 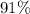 . (Rounded to two significant figures, as in other quantities in the question.)
. (Rounded to two significant figures, as in other quantities in the question.)
Explanation:
Start with the balanced chemical equation:
Look up relevant relative atomic mass data on a modern periodic table:
Calculate the formula mass of the species:
There are two reactants in this reaction: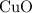 and
and 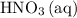 . Assume that
. Assume that 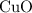 is the limiting one. In other words, assume that all the
is the limiting one. In other words, assume that all the 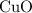 is consumed before
is consumed before 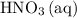 was.
was.
Consider: how many moles of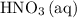 would be required to convert all that
would be required to convert all that 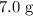 of
of 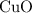 to
to 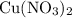 ?
?
Calculate the number of moles of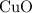 formula units in
formula units in  of
of
Note the ratio between the coefficients of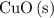 and
and 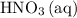 :
:
Therefore:
On the other hand, how many moles of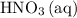 are actually available?
are actually available?
Convert the volume of that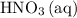 solution to the standard unit (liter.)
solution to the standard unit (liter.)
Calculate the number of moles of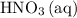 in that
in that 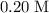 solution:
solution:
Apparently, the quantity of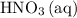 required exceeded the quantity that is available. Therefore, the assumption is invalid, and
required exceeded the quantity that is available. Therefore, the assumption is invalid, and 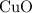 cannot be the limiting reactant. At the same time,
cannot be the limiting reactant. At the same time,
Mass of the reactant in excessSince it is now known that all that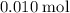 of
of 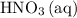 will be consumed, apply that coefficient ratio again to obtain the quantity of
will be consumed, apply that coefficient ratio again to obtain the quantity of 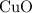 consumed in this reaction:
consumed in this reaction:
It was already shown that the formula mass of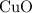 is (approximately)
is (approximately) 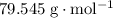 . Therefore, the mass of that
. Therefore, the mass of that 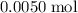 formula units of
formula units of 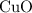 would be:
would be:
Before the reaction,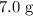 of
of 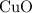 is available. Therefore, all that
is available. Therefore, all that 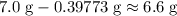 of
of 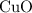 would be in excess.
would be in excess.
Percentage YieldSimilarly:
Apply this ratio to find the theoretical yield of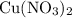 :
:
Find the mass of that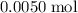 of
of 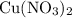 formula units using its formula mass:
formula units using its formula mass:
Calculate the percentage yield given that the actual yield is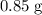 :
:
(Rounded to two significant figures.)
Ответ:
2Fecl
2Na2(OH)2
AlSO4 + H
Explanation:
metal and salt gives a metalsalt and gas