A river is contaminated with 0.65 mg/L of dichloroethylene (C2H2Cl2). What is the concentration (in ng/L) of dichloroethylene at 21°C in the air breathed by a person swimming in the river (kH for C2H2Cl2 in water is 0.033 mol/L•atm)?
Solved
Show answers
More tips
- F Food and Cooking How to Make Cottage Cheese at Home: Simple and Quick Recipe with Step By Step Instructions...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
Answers on questions: Chemistry
- C Chemistry AP chemistry, please explain how you got the answer if you can :( thank you so much...
- C Chemistry Which of these is an oxidation half-reaction?0 V3+ + e + V2+1 Fe + 02 - Fe203o c4+ + 2e → C2+...
- C Chemistry Which pair of compounds will form a buffer in aqueous solution? hcn and hcl nacn and naoh hcl and naoh nacn and kcn hcn and nacn hcl and nacl?...
- C Chemistry If the oxidation state of h is +1 and o is −2, what is the oxidation state of c in c2h4o? −1 +2 +1 −2...
- C Chemistry A truck with a mass of 30,000 kg is traveling down a Michigan Highway at a speed of 32 m/s when it sees a roadblock and must come to a sudden stop in 13 seconds. Given a...
- C Chemistry Name the following ionic compound: Ba(OH)2*2H2O...
- C Chemistry The chemical equation below shows the reaction between tin (Sn) and hydrogen fluoride (HF). Sn + 2HF Right arrow. SnF2 + H2 The molar mass of HF is 20.01 g/mol. How many...
- M Mathematics Using the exxon data as an example, pretend penny’s pickles (a fake pickle company) divides their company into 10,000 shares of stock. if you buy 200 of those shares, what...
- H Health I ma guy who wants to grow out his haor like a girl. what so i need to do?...
- H History The idea of supply and demand is based on the development of a. business decisions in a thriving economy. b. sufficient products to meet consumer wants. c. governmental...

Ответ:
The concentration of dichloroethylene in the air breather by a person swimming in the river is
Explanation:
We are given:
Concentration of dichloroethylene = 0.65 mg/L
Converting this into mol/L, we use the conversion factor:
1 g = 1000 mg
Molar mass of dichloroethylene = 97 g/mol
Converting the concentration, we get:
To calculate the pressure of the gas, we use ideal gas equation:
where,
P = pressure of the gas
C = concentration of gas =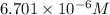
R = Gas constant =
T = temperature of the gas =![21^oC=[21+273]K=294K](/tpl/images/0561/0483/32d52.png)
Putting values in above equation, we get:
To calculate the concentration of gas we use the equation given by Henry's law, which is:
where,
Putting values in above equation, we get:
Converting this into ng/L, we use the conversion factor:
Molar mass of dichloroethylene = 97 g/mol
Converting the concentration, we get:
Hence, the concentration of dichloroethylene in the air breather by a person swimming in the river is
Ответ:
3.55 × 10n M n = -10