cseiver420
22.04.2020 •
Chemistry
A sample of air contains 78.08% nitrogen, 20.94% oxygen, 0.0500% carbon dioxide, and 0.930% argon by volume. How many molecules of each gas are present in 6.51 L of the sample at 43°C and 1.59 atm? Enter your answers in scientific notation.
Solved
Show answers
More tips
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- F Family and Home What is Most Important in Men s Lives?...
- G Goods and services Which TV is better - LCD or Plasma?...
- C Computers and Internet Are there special gaming mice?...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
- C Computers and Internet 3D Glasses! What is this thing?...
Answers on questions: Chemistry
- C Chemistry Hello guys, i am a gcse student and i was wondering if any of you have or know where to find the gcse aqa 2020 chemistry paper 2 for combined science that would...
- C Chemistry How is matter conserved on a molecular scale when ionic compounds dissociate in solution?...
- C Chemistry A scientist is studying a protist and records the following information about it: Cell has a nucleus Has a flagella Produces oxygen Found in salt water Which conclusion...
- C Chemistry How does the celsius thermometer work...
- C Chemistry On a cool morning, uyen’s breath can form a cloud when she breathes out. which changes of state are most responsible for uyen seeing her breath in this way? melting...
- C Chemistry The layer of earth s atmosphere where atoms and molecules escape into space is referred to as the...
- C Chemistry What is true of a basic solution at room temperature? it has a ph value below 7. it has a higher concentration of hydroxide ions than hydronium ions. it has a distinct...
- C Chemistry Which one is better jolly ranchers or starbursts...
- M Mathematics How many 0.45m are there in 8.1m...
- M Mathematics If P = 7, what is 76 - (13 + 2p) ?...

Ответ:
Answer : The number of molecules of nitrogen, oxygen, carbon dioxide and argon is, ,
,  ,
,  and
and  respectively.
respectively.
Explanation :
First we have to calculate the total moles of mixture of gas by using ideal gas equation.
where,
P = Pressure of mixture of gas = 1.59 atm
V = Volume of mixture of gas = 6.51 L
n = number of moles mixture of gas = ?
R = Gas constant =
T = Temperature of mixture of gas =
Putting values in above equation, we get:
Now we have to calculate the moles of nitrogen, oxygen, carbon dioxide and argon.
Moles of nitrogen =
Moles of oxygen =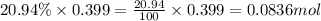
Moles of carbon dioxide =
Moles of argon =
Now we have to calculate the number of molecules of nitrogen, oxygen, carbon dioxide and argon.
As, 1 mole of nitrogen contains number of molecules of nitrogen.
number of molecules of nitrogen.
So, 0.312 mole of nitrogen contains number of molecules of nitrogen.
number of molecules of nitrogen.
and,
As, 1 mole of oxygen contains number of molecules of oxygen.
number of molecules of oxygen.
So, 0.0836 mole of oxygen contains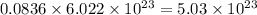 number of molecules of oxygen.
number of molecules of oxygen.
and,
As, 1 mole of carbon dioxide contains number of molecules of carbon dioxide.
number of molecules of carbon dioxide.
So, 0.000199 mole of carbon dioxide contains number of molecules of carbon dioxide.
number of molecules of carbon dioxide.
and,
As, 1 mole of argon contains number of molecules of argon.
number of molecules of argon.
So, 0.00371 mole of argon contains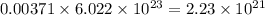 number of molecules of argon.
number of molecules of argon.
Therefore, the number of molecules of nitrogen, oxygen, carbon dioxide and argon is, ,
,  ,
,  and
and  respectively.
respectively.
Ответ:
The italicized portion of the sentence "looking for unusual flowers" is a present participle phrase ("looking" is the verb in present participle) used after a verb of movent, in this case "walk".
It is used to indicate parallel activity: in this case as they were walking along the mountain path, they were looking for unusual flowers.
The structure is: present participle (looking for) and the object (flowers) which is pre modified by the adjective unusual.