A sample of neon occupies a volume of 478 mL at STP. What will be the volume of the neon when the pressure is reduced to 93.3 kPa?
Solved
Show answers
More tips
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- D Dating, Love, Relationships How Long Can Love Last?...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- A Auto and Moto Discovering the Leader: What is the Most Expensive Car in the World?...
- F Food and Cooking How to Quickly Put your Child to Sleep?...
- C Computers and Internet How to Create a Website for Free and Easy?...
Answers on questions: Chemistry
- C Chemistry 1350 mL of gas at standard temperature and pressure is compressed to 734 ml. What is the new pressure of the gas? a) 0.55 atm b) 1.84 atm c) 5.58 atm d) 11.8 atm...
- C Chemistry What is the density of Iodine gas at 2.36 atm and 65.0 °C? A) 0.026 g/L B) 0.046 g/L C) 21.6 g/L D) 38.3 g/L...
- C Chemistry Calculate the volume of 2M sulfuric acid that must be diluted with water to produce 2dm^3 of 0.5M sulfuric acid....
- C Chemistry Volume of 4.3 moles of CO2...
- C Chemistry Number of moles In 52.3 g of C12H22O11...
- C Chemistry What is different between focus of an earthquake and the epicenter of an earthquake?...
- C Chemistry 1. Indique si los siguientes enunciados son falsos (F) o verdaderos (V), según corresponda: - Una corriente eléctrica es un flujo de electrones que circulan a través...
- C Chemistry FIRST TO ANSWER CORRECTLY GETS BRAINELSIT! pls asap :)...
- C Chemistry What surprised you about the ways Red, Green, and Blue light interacted to create different colors?...
- C Chemistry Pure copper coin has a mass of 3.25g how many moles of copper are in the coin...

Ответ:
The volume of neon when the pressure is reduced to 93.3 kPa is 519 mL.
Explanation:
The kinetic theory of gases is mostly based on Boyle's law. From the Boyle's law, the pressure experienced by any gas molecules is inversely proportional to volume of the gas molecules. Also this inverse relation is obeyed if and only if the number of moles and temperature of the gas molecules remained constant.
So,
So if there is a change in pressure then there will be inverse change in volume. That means if there is decrease in the pressure of gas molecules then there will be increase in the volume and vice versa.
So the Boyle's law is combined as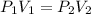
As here the initial pressure or is 1 atm or 101.3 kPa and the initial volume is 478 mL. Similarly, the final pressure is 93.3 kPa and the final volume will be
is 1 atm or 101.3 kPa and the initial volume is 478 mL. Similarly, the final pressure is 93.3 kPa and the final volume will be
So, the volume of neon when the pressure is reduced to 93.3 kPa is 519 mL.
Ответ:
begins in the mouth