A student titrates an unknown weak acid, HA, to a pale pink phenolphthalein endpoint with 25.0 mL of 0.100 M NaOH. The student then titrates in 11.0 mL of 0.100 M HCl to the solution containing the neutralized unknown acid (HCl was delivered from a second buret). The pH of the resulting solution is pH
Solved
Show answers
More tips
- H Health and Medicine Impeccable Memory: How to Improve It...
- L Leisure and Entertainment How Many Seasons are There in the TV Show Interns?...
- S Sport When will the Biathlon World Championships 2011 take place in Khanty-Mansiysk? Answers to frequently asked questions...
- H Health and Medicine Trading Semen for Money: Where Can You Sell and Why Would You Want to?...
- F Food and Cooking Homemade French Fries: The Ultimate Guide...
- H Health and Medicine How to Increase Blood Pressure without Medication?...
- S Style and Beauty Choosing a Hair Straightener: Specific Criteria to Consider...
- F Food and Cooking How to Make Polendwitsa at Home?...
- S Science and Technology When do we change our clocks?...
- L Leisure and Entertainment What to Give a Girl on March 8?...
Answers on questions: Chemistry
- C Chemistry Which atom had the largest ionization energy 1) Na 2)cl 3)O 4)Se...
- C Chemistry Put in order from least to most: Electronegativity A) o b)F c)Li d) B e) C...
- C Chemistry Plzzz help me out as I was not in class and I missed the lesson...
- C Chemistry 3) Which pair of formulas represents the empirical formula and molecular formula for a certain (the same) compound? A. CH4 and C2H6 B. CH2O and C4H6O C. O2 and O3 D. C3H403...
- C Chemistry What is the pressure exerted by 0.122 mol oxygen gas in a 1.50-L in a container at room temperature...
- C Chemistry Campers want a travel cup that uses a cold pack to cool a liquid. Design the outer part of the cup that keeps the drink cold once it has been cooled down. me no have Materials...
- C Chemistry 26. Calculate the mass of hydrogen formed when 25 grams of aluminum reacts with excess hydrochloride acid. 2AI + 6HCl →l Al2Cl6 + 3H2 ...
- C Chemistry What the recipe of momo...
- C Chemistry Calculate the percentage of O in CH₃COOH...
- C Chemistry What is the molecular formula of a compound that is 51.02 % carbon, 13.80 % hydrogen, and 35.18 % oxygen? The molecular mass is 88.0 g/mol....

Ответ:
Hello your question is incomplete the pH of the solution is unknown hence I will take the pH of the solution be = 4.7
answer : Pka = 5 i.e. > 4.7
Explanation:
Student titrates :
Unknown weak acid ( HA ) with 25 mL of 0.100 M of NaOH ( to a pale pink phenolphthalein endpoint ) then titrates with 11.0 mL of 0.100 M HCL to the solution that contains neutralized unknown acid
Assuming pH of solution = 4.7
HA + NaOH --------> H2O + NaA
at equilibrium point
0.1 * 25 * 1 = n * 1 ∴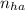 = 2.5 mmoles ------ 1
= 2.5 mmoles ------ 1
also
at equilibrium point
The ratio of reaction 2 to 1
Determine the pKa value of the solution
Given that
pH = Pka + log ( A^- / hA )
4.7 = Pka + log ( 1/2 )
therefore Pka = 4.7 - ( -0.3 ) = 5
Ответ:
Hi
Explanation: