A0.642g sample of an unknown gas was collected over water at 25.0°c and 1.04 atm. the collection cylinder contained 151.3 ml of gas after the sample was released. find the molar mass of the unknown gas.
Solved
Show answers
More tips
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry Please help :( How much NaOH (in moles) must be added to 1L of a buffer solution that is 1.8 M in acetic acid and 1.2 M in sodium acetate to result in a buffer solution...
- C Chemistry Which of the following bond has highest bond energy? A.O=OB.H-CLC.C=C D.H-H...
- C Chemistry Why are radio waves good for transmitting information long distances?...
- C Chemistry What is the electronic structure of x-2...
- C Chemistry 2CO + O2 2CO2 How many liters of carbon dioxide are produced if 75 liters of carbon monoxide are burned in oxygen? How many liters of oxygen are necessary?...
- C Chemistry What is the name of the warm phase associated with melting ice and higher sea level? PL ILL GIVE 5 STARS PLSSS JUST PLS HELP ME ITS FOR MY MIDTERM REVIEW ;-; ITS DUE...
- C Chemistry How many bonds can sodium form...
- C Chemistry Which of the materials in the chart originally came from fossil fuels? Group of answer choices Acrylic Cotton Nylon Polyester Viscose...
- C Chemistry Need helpK2O+HNO3›K2NO3+H2O balance...
- M Mathematics In science, students are observing the growth of to plants. Plant A receives suntight and Plant B does not The graph and table show the data for this scenario What...

Ответ:
Answer : The molar mass of unknown gas is 99.8 g/mole.
Explanation :
Using ideal gas equation:
where,
P = pressure of gas = 1.04 atm
V = volume of gas =
conversion used :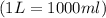
T = temperature of gas =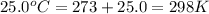
R = gas constant = 0.0821 L.atm/mole.K
w = mass of an unknown gas = 0.642 g
M = molar mass of an unknown gas = ?
Now put all the given values in the ideal gas equation, we get:
Therefore, the molar mass of unknown gas is 99.8 g/mole.
Ответ: