rainbow7020
26.08.2019 •
Chemistry
A24.0 g sample of nitrogen gas reacts with an excess of hydrogen gas to give an actual yield of 3.85 g nh3. what is the percent yield for this reaction given the reaction: n2(
g. 3h2(
g. --> 2nh3(g
Solved
Show answers
More tips
- F Family and Home When and how to start introducing solid foods to your baby?...
- B Business and Finance Moneybookers – What it is and How it Works...
- C Computers and Internet How to Format Your C Drive: Detailed Guide and Tips...
- F Food and Cooking What can and cannot be eaten during Lent?...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- D Dating, Love, Relationships Is it a Compliment or Flattery: What s the Difference?...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- C Computers and Internet What to Do If Your ICQ Gets Hacked?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry Complete the chemical equation. Which numbers belong in the blanks? Your answer should contain 4 numbers listed in order (ex: 1, 2, 3, 4) NEED HELP ASAP...
- C Chemistry How are photosynthesis and cellular respiration related? They have opposite equations O They have the same equation O They both produce carbon dioxide O They both produce oxygen...
- C Chemistry In the Seventhgroup, what isthe type of bond that is likely toformadaThegroup 7 element react vigorouely with group a element such as sulromand potassium in each cause, a metal...
- C Chemistry Calculate the mass, in grams, of 0.433 mole of calcium nitrate...
- C Chemistry What is the difference between a seismograph and seismogram? a) a seismograph is an instrument that records movements within the earth. a seismogram is an instrument that monitors...
- C Chemistry The quantum mechanical model of the atom a) is concerned with the probability of finding an electron in a certain position b) has many analogies in the visible world c) defines...
- C Chemistry Is this statement true or false? you can make an iron nail magnetic by wrapping it in wire and sending electricity through the wire. a. true b. false...
- C Chemistry Use the drop-down menus to identify the class to which the named compounds belong. 2-heptanol, triethylamine, benzoic acid, decanal...
- C Chemistry Achemical company makes two brands of antifreeze. the first brand is 70% pure antifreeze, and the second brand is 95% pure antifreeze. in order to obtain 160 gallons of a mixture...
- C Chemistry The half-life of na-24 is 15 hours . when there are 1000 atoms of na-24 in a sample , a scientist starts a stopwatch . the scientist stops the stopwatch at 45 hours , there are...

Ответ:
6.62%
Explanation:
We have the following reaction
A real yield of 3.85 g of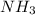 is obtained
is obtained
To calculate the reaction yield in percentage we must calculate the mass that theoretically we must obtain
The reaction occurs in excess of hydrogen and 24.0g of N this means that when the nitrogen is finished the reaction will end
Nitrogen is our limit reagent
Molar mass of nitrogen
This means that in 1 mol of nitrogen there is 14 g.
To calculate the moles of nitrogen in 24 g N we apply a simple rule of three
According to stoichiometric coefficients 1 mol of N produces 2 moles of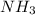
1.71 mol N how many moles of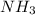 will produce
will produce
Molar mass of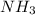 17 g / mol
17 g / mol
This means that in 1 mol of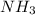 there are 17 g.
there are 17 g.
58.14g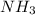 corresponds to the theoretical yield of the reaction
corresponds to the theoretical yield of the reaction
To calculate the percentage yield of the reaction we use the following formula
The percent yield for this reaction is 6.62%
Ответ:
He ripped his other suit