A51.24-g sample of ba(oh)2 is dissolved in enough water to make 1.20 l of solution. how many milliliters of this solution must be diluted with water in order to make 1.00 l of 0.100 m ba(oh)2?
Solved
Show answers
More tips
- A Auto and Moto Which alarm system to choose?...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- F Food and Cooking How many stages of coffee roasting are there?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- F Food and Cooking Experts Name Top 5 Healthiest Teas...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Liver Cleansing - Rejuvenation for the Body?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- S Style and Beauty Unbelievable Hairstyles for Long Hair - 2021 Trends...
Answers on questions: Chemistry
- H History Since George Washington’s warning in his 1796 Farewell Address that the United States should stay out of European Alliances, the United States had followed a policy...
- C Chemistry 1. Communicate Explain how you would calculate the wavelength of a wave if you knew both the wave s speed and frequency. Type your answevere...
- H History What were the black codes and how did they limit the rights of african americans in the south...

Ответ:
361.5 ml of water will required to make 1.00 L of 0.100 M .
.
Explanation:
Mass of barium hydroxide = 51.24 g
Volume of solution = 1.20 L
= 1.20 L
Molarity of the solution =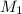
Let the volume of the solution 0.2766 M to be diluted be
Desired concentration of barium hydroxide solution =
Volume of the 0.1000 M solution =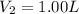
361.5 ml of water will required to make 1.00 L of 0.100 M .
.
Ответ:
Pi is your initial amount (120 grams)
0.5 as your exponential base represents your HALF life hence 0.5
t represents the time elapsed
c represents your given half life value of (5730)
P(t) represents the exponential function of your half life
120/2 =60
60/2 = 30
30 /2 = 15
As you can see above, 3 half lives of 5730 years EACH degraded the carbon-14 to 15 grams. You divide each number by two as it is a HALF life.
P(t)= Pi (0.5) ^t/c
P(17190) = 120 (0.5) ^17190/5730
P(17190) = 120 (0.5) ^3
P(17190) = 15 grams
So your answers should be 3 half lives or after 17190 years have elapsed!