foziasif724
29.08.2019 •
Chemistry
Abuffer is prepared by combining 25 ml of 0.5o mnh(aq) with 25 ml of 0.20 m hci. what is the ph of the buffer? (ka (nh4) = 5.6 x 10 ) a. 8.86 b. 9.65 c. 8.00 d. 7.76 e. 9.43
Solved
Show answers
More tips
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry When a reaction takes place in a sealed flask, the total mass of the...
- C Chemistry Does a substance that evaporates more quickly (thus giving a greater δt o c) have a higher or lower force of attraction to other molecules in the liquid compared...
- C Chemistry What volume of O2 at STP is required to oxidize 8.0 L of NO at STP to NO2? What volume of NO2 is produced at STP?...
- C Chemistry Molecular mass expressed in gram is called...
- C Chemistry Some factories have started using large tanks of bacteria to remove carbon dioxide from the atmosphere. As more factories start to do this, the amount of carbon...
- C Chemistry What is the mass of a liquid that has a volume of 50.0ml and a density of 2.40g/ml?...
- C Chemistry Given that D is the midpoint of AB and K is the midpoint of BC, which statement must be true?...
- H History Which form of dissent and protest is represented when you refuse to be a customer of a company?...
- E English How do you believe ballet is an authentic portrait of movement?...
- P Physics E 3.6 What force is needed to give a mass of 20 kg an acceleration of 5 m/s??...

Ответ:
e) 9.43
Explanation: A buffer solution is a mixture of a weak acid and its conjugate base or a weak base and its conjugate acid and the pH of the buffer is calculated using Handerson equation:
There are 25 mL of 0.50 M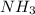 which is a weak base. 25 mL of 0.20 M HCl, a strong acid are added. Ammonia reacts with HCl to form its conjugate acid, ammonium ion. The net ionic equation will be:
which is a weak base. 25 mL of 0.20 M HCl, a strong acid are added. Ammonia reacts with HCl to form its conjugate acid, ammonium ion. The net ionic equation will be:
From above reaction, ammonia and HCl react in 1:1 mol ratio. Let's calculate the moles of each we have before the reaction. There is also, 1:1 mol ratio between HCl and ammonium ion.
moles of ammonia =
= 0.0125 mol
moles of HCl =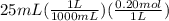
= 0.005 mol
Excess moles of ammonia = 0.0125 - 0.005 = 0.0075 mol
moles of ammonium ion formed = 0.005 mol
Total volume of the solution = 0.025 mL + 0.025 mL = 0.050 L
concentration of ammonia in buffer =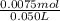
= 0.15 M
concentration of ammonium ion in buffer =
= 0.10 M
pKa is calculated from given Ka as:
pKa = 9.25
Plug in the values in Handerson equation:
pH = 9.25 + 0.18
pH = 9.43
So, the correct choice is e) 9.43 .
Ответ:
The [H⁺] of the solution is 1.74*10⁻⁹ M
Explanation:
pH, short for Hydrogen Potential, is a measure of acidity or alkalinity that indicates the amount of hydrogen ions present in a solution or substance. The pH scale ranges from 0 to 14. Values less than 7 indicate the acidity range and those greater than 7 indicate alkalinity or basicity. Value 7 is considered neutral.
The pH is calculated as the negative logarithm (base 10) of the molar concentration of the hydrogen or proton ions (H⁺) or hydronium ions (H₃O⁺).
pH= - log [H⁺] = -log [H₃O⁺]
In this case, you know that pH= 8.76. Replacing in the definition of pH:
8.76 = -log [H⁺]
Solving:
[H⁺]= 10⁽⁻⁸ ⁷⁶⁾
[H⁺]= 1.74*10⁻⁹ M
The [H⁺] of the solution is 1.74*10⁻⁹ M