gabbym39077
26.12.2019 •
Chemistry
Acetic acid (ch3cooh) is formed from its elements by the following reaction equation:
2 c (graphite) + 2 h2 (g) + o2 (g) → ch3cooh (g)1. what is the volume of acetic acid ch3cooh gas produced by the reaction of 18.6 grams of h2 gas at 35°c and 1.05 atm?
Solved
Show answers
More tips
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- C Chemistry True or false during reactions or light is needed carbon dioxide is converted to glucose...
- C Chemistry LiOH + KCI → LiCl + KOHa) I began this reaction with 20.0 grams of LiOH. What is my theoretical yield of LiCI?...
- C Chemistry Which two categories can all substance be classified?...
- C Chemistry 4HCI + O2 =2H20 + Cl2 Which of the following would result from increasing the concentration of H20? The rate of the backward reaction increases. The volume of hydrochloric...
- C Chemistry Matilda is able to get her brother s hair to move toward the balloon without touching his hair because...
- C Chemistry HELPPP Fill the missing blanks...
- C Chemistry Fill in for the missing blacks...
- C Chemistry How many liters of O2 are present in 175 grams O2? (32.00 grams O2 per 1 mole Oz, also 22.4 L 02 per 1 mole Oz)...
- C Chemistry What does the trapdoor spider do if the availability of food and water was limited?...
- C Chemistry Which of the following gases would have the greatest volume at stp? . a. 5.00 moles of h2 gas. b. 5.00 g of rn gas. c. 6.02 × 1023 particles of ne gas. d. all of the...

Ответ:
111 L
Explanation:
Calculation of moles of hydrogen gas:-
Mass of = 18.6 g
= 18.6 g
Molar mass of = 2.01588 g/mol
= 2.01588 g/mol
According to the given reaction:-
2 moles of hydrogen gas on reaction produces one mole of acetic acid gas.
So,
1 mole of hydrogen gas on reaction produces mole of acetic acid gas.
mole of acetic acid gas.
Also,
9.23 mole of hydrogen gas on reaction produces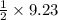 mole of acetic acid gas.
mole of acetic acid gas.
Moles of acetic acid gas = 4.615 moles
Given that:
Temperature = 35 °C
The conversion of T( °C) to T(K) is shown below:
T(K) = T( °C) + 273.15
So,
T = (35 + 273.15) K = 308.15 K
n = 4.615 moles
P = 1.05 atm
V = ?
Using ideal gas equation as:
where,
P is the pressure
V is the volume
n is the number of moles
T is the temperature
R is Gas constant having value = 0.0821 L atm/ K mol
Applying the equation as:
1.05 atm × V = 4.615 moles ×0.0821 L atm/ K mol × 308.15 K
⇒V = 111 L
Ответ:
The correct answer is The 19th amendment barred voting discrimination based on sex.
In the question it refers to citizenship. In the United States, citizenship refers to the ability of an individual to participate in American politics. Knowing this, you can see that expanding this type of right would mean that more people have the ability to vote. The only answer that deals with more people getting the right to vote is letter choice B.