williamlindley2
26.06.2019 •
Chemistry
Acompound is decomposed by an electric current giving 19.98 % g o and 2.52 g h what is the % composition of this compound. (find the % by mass of each element)
Solved
Show answers
More tips
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry My teacher says protons and electrons are always equal to each other. So does that mean every element has a charge of 0? No, that can’t be.. but I’m still confused. Please help!...
- B Business Refers to cross-checking the transactions you logged in your checkbook register with your...
- E English Which of the following is a primary difference between a short story and a novel?...
- E English If i take 6-8 pills of exedrin rn, what will happen!?...
- C Computers and Technology How to format each dictionary item as a text string in the input file. How to covert each input string into a dictionary item. How to format each item of your inverted dictionary...
- M Mathematics Im POSSIBLE POINTS: 5.56 1. Lance applied to work for two different food-delivery companies. One company pays their drivers $500 per week plus $0.60 per mile they drive. The...

Ответ:
We have to calculate the percentage composition of the compound.
The answer is: amount (in mass percentage) of oxygen is 88.8 % and amount (in mass percentage) of hydrogen is 11.2 %.
We know atomic mass of oxygen is 16 and atomic mass of hydrogen is 1.
The amount of Oxygen produced= 19.98 g and amount of Hydrogen produced= 2.52 g. So, total amount of the compound is: (19.98 + 2.52) g= 22.50 g.
So, mass percentage of oxygen is (Mass of oxygen produced/ total mass of compound) X 100 =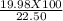 = 88.8 %
= 88.8 %
Masspercentage of hydrogen is: (Mass of hydrogen produced/ total mass of compound) X 100 = = 11.2 %.
= 11.2 %.
Ответ:
i wish i could but i just dont know this...
Explanation: