Aluminum reacts with excess hydrochloric acid to form aqueous aluminum chloride and 46.5 mL of hydrogen gas over water at 27 degrees Celsius and 751 mmHg. How many grams of aluminum reacted? The partial pressure of water at 27 C is 26.8 mmHg.
Solved
Show answers
More tips
- H Health and Medicine How to Improve Eyesight: Science-based Techniques to Enhance Your Visual Acuity...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- H Health and Medicine Tick Traps: How to Remove Them Safely and Effectively...
- S Society and Politics Why are thugs called gopniks ? A fascinating journey through Russian subculture...
- A Animals and plants Want a Perfect Lawn? Learn How to Plant Grass the Right Way...
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...

Ответ:
The amount of aluminium reacted is 0.0324 grams
Explanation:
We are given:
Vapor pressure of water = 26.8 mmHg
Total vapor pressure = 751 mmHg
Vapor pressure of hydrogen gas = Total vapor pressure - Vapor pressure of water = (751- 26.8) mmHg = 724.2 mmHg
To calculate the amount of hydrogen gas collected, we use the equation given by ideal gas which follows:
where,
P = pressure of the gas = 724.2 mmHg
V = Volume of the gas = 46.5 mL = 0.0465 L (Conversion factor: 1 L = 1000 mL)
T = Temperature of the gas =![27^oC=[27+273]K=300K](/tpl/images/0537/7176/16c81.png)
R = Gas constant =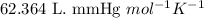
n = number of moles of hydrogen gas = ?
Putting values in above equation, we get:
The chemical equation for the reaction of aluminium with HCl follows:
By Stoichiometry of the reaction:
3 moles of hydrogen gas is produced when 2 moles of aluminium is reacted
So, 0.0018 moles of hydrogen gas will be produces when =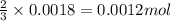 of aluminium is reacted
of aluminium is reacted
To calculate the mass from given number of moles, we use the equation:
Moles of aluminium = 0.0012 moles
Molar mass of aluminium = 27 g/mol
Putting values in above equation, we get:
Hence, the amount of aluminium reacted is 0.0324 grams
Ответ:
it is a chemical change
explanation: