janaes1217
19.05.2021 •
Chemistry
An arctic weather balloon is filled with 38.5 L of helium gas inside a prep shed. The temperature inside the shed is 8. °C. The balloon is then taken outside, where the temperature is -41. °C. Calculate the new volume of the balloon. You may assume the pressure on the balloon stays constant at exactly 1 atm. Be sure your answer has the correct number of significant digits.
Solved
Show answers
More tips
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- D Dating, Love, Relationships How Long Can Love Last?...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- A Auto and Moto Discovering the Leader: What is the Most Expensive Car in the World?...
- F Food and Cooking How to Quickly Put your Child to Sleep?...
- C Computers and Internet How to Create a Website for Free and Easy?...
- F Family and Home Parquet or laminate, which is better?...
Answers on questions: Chemistry
- C Chemistry Part A Measure and record the masses of all the metal strips you set out in front of the test tubes....
- M Mathematics Pls help :) A circle centered at (0,-8) passes through the point (2,-5) Find the equation of the circle. Write your equation in the form (x - h)^2 + (y - k)^2 = r^2...
- P Physics 1. What is the equivalent resistance of the circuit?...
- P Physics What is the charge on a hypothetical ion with 55 protons and 52 electrons?...

Ответ:
The new volume of the balloon is 197.31 L.
Explanation:
Given: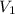 = 38.5 L,
= 38.5 L, 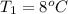
According to Charles law, at constant pressure the volume of a gas is directly proportional to temperature.
Formula used to calculate the new volume is as follows.
Substitute the values into above formula as follows.
Thus, we can conclude that the new volume of the balloon is 197.31 L.
Ответ:
For 1 mole of a single atom it is equal to its molar mass. And a single atom, 1 mole is equal to the Avogadro's Number.
Explanation: The relationship can be expressed through the following:
1 mole = molar mass of an atom/ compound
1 atom x 1 mole / 6.022x10^23 atoms