bahjaMcclatchey1110
25.01.2020 •
Chemistry
An electron is confi ned to a linear region with a length of the same order as the diameter of an atom (about 100 pm). calculate the minimum uncertainties in its position and speed.
Solved
Show answers
More tips
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- O Other Childhood Fears: What Many of Us Experienced...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
Answers on questions: Chemistry
- E English What kind of clause is the clause in dark type? i do not know whether everyone will be on time. a. adjective clause b. adverb clause c. noun clause d. independent clause...
- B Biology What is ur favorite zodiac sign other than ur own?...
- M Mathematics What value of x makes the equation true 4x = 32?...
- B Biology help please the link above ignore picture...

Ответ:
Explanation:
The minimum uncertainty position is 100 nm.
Therefore, by Heisenberg Uncertainty principal
ΔxΔp≥h/2
Δp≥h/(2Δx)
we know that
h=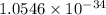 Js
Js
Δx=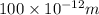
therefore,
therefore,
Ответ:
li = 2
S = 1
O = 4
= 7