An unknown solid is placed on a scale and has a mass of 2.5 grams. The solid then displaces 35 mL of water in a graduated cylinder. Find the unknown solid’s density.
Solved
Show answers
More tips
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...

Ответ:
The unknown solid’s density is 0.071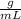
Explanation:
Density is defined as the property that matter, whether solid, liquid or gas, has to compress into a given space. In other words, density is a quantity that allows us to measure the amount of mass in a certain volume of a substance. Then, the expression for the calculation of density is the quotient between the mass of a body and the volume it occupies:
From this expression it can be deduced that density is inversely proportional to volume: the smaller the volume occupied by a given mass, the higher the density.
Archimedes' principle states that every body immersed in a fluid experiences a vertical and upward thrust equal to the weight of fluid dislodged. In other words, this principle says that the volume of the object is equal to the volume of the water displaced or displaced. So:
mass= 2.5 gramsvolume=35 mLReplacing in the definition of density:
you get:
The unknown solid’s density is 0.071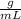
Ответ:
Answer : The volume of produced are 90.7 liters.
produced are 90.7 liters.
Explanation :
The balanced chemical reaction will be:
First we have to calculate the moles of PbS.
Molar mass of PbS = 239.26 g/mole
Now we have to calculate the moles of by using ideal gas equation.
by using ideal gas equation.
Using ideal gas equation :
where,
P = Pressure of gas = 2.00 atm
gas = 2.00 atm
V = Volume of gas = 123 L
gas = 123 L
n = number of moles = ?
= ?
R = Gas constant =
T = Temperature of gas =
gas = 
Putting values in above equation, we get:
The number of moles of is, 6.07 mole
is, 6.07 mole
Now we have to calculate the limiting and excess reagent.
From the balanced reaction we conclude that
As, 3 mole of react with 2 mole of
react with 2 mole of 
So, 6.07 moles of react with
react with 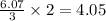 moles of
moles of 
From this we conclude that, is an excess reagent because the given moles are greater than the required moles and
is an excess reagent because the given moles are greater than the required moles and  is a limiting reagent and it limits the formation of product.
is a limiting reagent and it limits the formation of product.
Now we have to calculate the moles of
From the reaction, we conclude that
As, 3 mole of react to give 2 mole of
react to give 2 mole of 
So, 6.07 moles of react to give
react to give 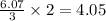 moles of
moles of 
Now we have to calculate the volume of produced at STP.
produced at STP.
As, 1 mole of contains 22.4 L volume of
contains 22.4 L volume of 
So, 4.05 mole of contains
contains  volume of
volume of 
Therefore, the volume of produced are 90.7 liters.
produced are 90.7 liters.