lucarignot
05.11.2020 •
Chemistry
Any help for this question ?

Solved
Show answers
More tips
- C Computers and Internet How to Download Movies from Torrents?...
- F Food and Cooking How to Make the Perfect Glühwein: Step-by-Step Guide...
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
Answers on questions: Chemistry
- C Chemistry A process creates a radioactive substance at the rate of 1 g/hr, and the substance decays at an hourly rate equal to 1/10 of the mass present (expressed in grams). Assuming...
- C Chemistry Short form of an element...
- C Chemistry Complete la siguiente información del cuadro...
- C Chemistry Given pOH = 3.7, what is the [H ?...
- C Chemistry Calculate the density of CF3Cl at 18 °C and a pressure of 114.0 kPa...
- C Chemistry List 10 testable questions for scientific Method...
- C Chemistry The a-helices and b- sheets in protein make up its...
- C Chemistry 1. How can she test this idea in the Flowing Water Model? I I...
- C Chemistry Which of the following does the atomic number of an atom represent? (1 point) o the number of neutrons in the atom O the number of particles outside the nucleus of the atom...
- C Chemistry Calculate with correct number of significant figures: 685.90 + 152.1 - 0.125...

Ответ:
Ответ:
Thus hydroxide ion concentration is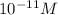
Explanation:
pH or pOH is the measure of acidity or alkalinity of a solution.
pH is calculated by taking negative logarithm of hydrogen ion concentration and pOH is calculated by taking negative logarithm of hydroxide ion concentration.
Given: pH=3
Thus hydroxide ion concentration is