memorybuxton
12.12.2019 •
Chemistry
Areaction vessel is charged with hydrogen iodide, which partially decomposes to molecular hydrogen and iodine: 2hi (g) h2(g) + i2(g) when the system comes to equilibrium at 425 °c, phi = 0.708 atm, and ph2 = pi2 the value of kp at this temperature is
Solved
Show answers
More tips
- T Travel and tourism How to Use a Compass: A Beginner s Guide...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- F Food and Cooking How many stages of coffee roasting are there?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- F Food and Cooking Experts Name Top 5 Healthiest Teas...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Liver Cleansing - Rejuvenation for the Body?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- S Style and Beauty Unbelievable Hairstyles for Long Hair - 2021 Trends...
Answers on questions: Chemistry
- C Chemistry A worker is getting ready to install solar panels on a roof. She will start by placing the solar panels in two stacks. The diagram above shows the solar panels...
- M Mathematics If Malik uses 16 peaches to make fruit salad, how many plums will he most likely use?...
- M Mathematics Option 1: Use the applet to construct circles for problems 1 & 2 OR 3 & 4. Take a screenshot and insert them into your student companion. 1. Circle A, with...
- B Business What is the definition of Gross Domestic Product (GDP)? A. an indicator used to measure the overall happiness of citizens based on their community involvement B....
- B Biology What kinds of geological features can be created by each type of relative motion?...

Ответ:
The value of the equilibrium constant: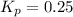
Explanation:
Given reaction: 2HI (g) ⇌ H₂(g) + I₂(g)
Number of moles of- HI: n₁ = 2 mole; H₂: n₂ = 1 mole; I₂: n₃ = 1 mole
Total number of moles: n = n₁ + n₂ + n₃ = 2 + 1 + 1 = 4 moles
The equilibrium constant for the given reaction is given as:
Given: Temperature: T = 425 °C = 425 + 273 = 698 K
The partial pressure: pHI = 0.708 atm,
and, pH₂ = pI₂
∵ partial pressure of a given gas: pₐ = Χₐ . P
Here, P is the total pressure
Χₐ is the mole fraction of given gas and is given by the equation
Mole fraction for HI: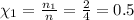
Mole fraction for H₂: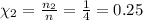
Mole fraction for I₂: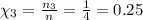
Thus, Χ₂ = Χ₃ = 0.25
The partial pressure of HI is given by;
pHI = Χ₁ P
0.708 atm = 0.5 × P
⇒ P = 1.416 atm
As the partial pressures: pH₂ = pI₂
∴ pH₂ = pI₂ = Χ₂ P = Χ₃ P = 0.25 × 1.416 atm = 0.354 atm
Therefore, the value of Kp can be calculated as:
Therefore, the value of the equilibrium constant: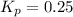
Ответ:
4.46 L
Explanation: By using Charles’s Law, we know that V1/T1=V2/T2. And whenever temperature is involved, you have to convert from Celsius to Kelvin (C° + 273). So, 4.00/ 24 +273 = V2/ 58 + 273. So the answer is, around 4.46 L.