aminsaad4253
20.01.2020 •
Chemistry
Asample of ch4 is confined in a water manometer. the temperature of the system is 30.0 °c and the atmospheric pressure is 98.70 kpa. what is the pressure of the methane gas, if the height of the water in the manometer is 30.0 mm higher on the confined gas side of the manometer than on the open to the atmosphere side. (density of hg is 13.534 g/ml).
Solved
Show answers
More tips
- F Food and Cooking Why Doesn t the Confirmation Link Come to Email?...
- L Leisure and Entertainment What Movies You Should Watch: A Guideline to Make the Right Decision...
- F Family and Home How to Choose a Baby Stroller: Tips from Experienced Parents...
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
Answers on questions: Chemistry
- B Biology Proteins are the building blocks of life. Every cell in the human body contains protein. Which organelles are likely to be abundant in cells such as fibroblasts that actively...
- S Spanish Ellas mucho en todas las tiendas. 1.mira 2.miran 3.miramos 4.miras...
- E English How many years older is Jem than his sister Scout?...
- S Social Studies Aperson’s behavior that might appear disordered if it occurred on a street corner in the united states, could also appear appropriate and understandable if it occurred in...
- M Mathematics Adam and brian are standing some distance apart on the side of a building 50 m tall .from where adam stands ,the angle of elevation of the top of the building is 30 from...

Ответ:
Explanation:
The given data is as follows.
T = = (30 + 273) K = 303 K
= (30 + 273) K = 303 K
height (h) = 30 mm = 0.03 m (as 1 m = 100 mm)
Density = 13.534 g/mL =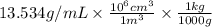
= 13534
The relation between pressure and atmospheric pressure is as follows.
P =
Putting the given values into the above formula as follows.
P =
=
= 102683.05 Pa
= 102.68 kPa
thus, we can conclude that the pressure of the given methane gas is 102.68 kPa.
Ответ:
B. mole/s
Explanation:
Molar mass is defined as the mass of 1 mole of the compound. It is calculated by adding the masses of all the elements present in the compound.
A mole is defined as the amount of substance that contains Avogardro number of the substance. Avogadro's number is given by .
.
According to avogadro's law, 1 mole of every substance occupies 22.4 L at STP and contains avogadro's number of particles. Standard condition of temperature (STP) is 273 K and atmospheric pressure is 1 atmosphere respectively.
of particles. Standard condition of temperature (STP) is 273 K and atmospheric pressure is 1 atmosphere respectively.
Pressure of the gas is defined as the force exerted by the particles on the walls of the container. It is expressed in various terms like 'mmHg', 'atm', 'kiloPascals' etc..