pheonixhowls
05.09.2019 •
Chemistry
Asolution is prepared by dissolving 31.9 g cesium chloride in 60.0 g water. the volume of the solution is 63.3 ml. calculate the molality of the cscl solution.
Solved
Show answers
More tips
- F Food and Cooking How to properly cook shrimp: tips from professionals...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
Answers on questions: Chemistry
- C Chemistry A student carried out a titration to find the exact volume of hydrochloric acid that would neutralise 25.0 cm3 of sodium hydroxide solution. NaOH + HCl → NaCl + H2O...
- C Chemistry How many moles of KCl are present in 95.3 mL of 2.10 M KCl...
- C Chemistry Calculate the ph value of the solution5.00 M NH3 Kb = 1.80 x 10^5-...
- C Chemistry Sulfur dioxide is produced from zinc sulfide when it reacts with oxygen. If the typical yield is 86.78%, what mass of SO2 should be expected if 4897 g of ZnS is used?...
- C Chemistry any help would be appreciated anyone plz help ASAP identify lewis acids and lewis bases from the following: AlCl₃ , Ag⁺ , CH₃ OH , CN⁻ , OH⁻ ,FeCl₃...
- C Chemistry What are the molecular formula of 20 elements...
- C Chemistry Match the following chemical and physical changes and properties, there will be two answers for each....
- C Chemistry Explain formation of covalent bonds when atoms share electrons....
- C Chemistry 3 Ethene burns in an excess of oxygen to form carbon dioxide and water.The equation for the reaction isC₂H4 & 30₂2002 + 2H2O.The relative molecular mass of ethene...
- C Chemistry Why is nitrogen dioxide a gas at room temperature...

Ответ:
The molality of the CsCl solution is 3.2 m.
Explanation:
Molality of a solution is defined as the number of moles of solute dissolved per kg of the solvent.
where,
n = moles of solute
moles of solute =
Now put all the given values in the formula of molarity, we get
Molality =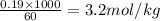
Thus molality of the CsCl solution is 3.2 m.
Ответ:
19kpa
Explanation:
The total pressure of a mixture of gases is the sum of the partial pressure exacted by the individual gases.
P total = P X + P Y
From the question above:
P total = P .nitrogen + P. oxygen
123.5kpa = 104.5kpa + P. Oxygen
P. Oxygen = 123.5kpa - 104.5kpa
= 19kpa