ramosramos142
20.01.2020 •
Chemistry
Asolution of chloroform, chcl3, and acetone, (ch3)2co, exhibits a negative deviation from raoult's law. this result implies that
Solved
Show answers
More tips
- F Family and Home Is it Worth Knowing the Gender of Your Child Before Birth?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
Answers on questions: Chemistry
- C Chemistry Cellular respiration uses glucose and oxygen to make...
- C Chemistry Calculate the kinetic energy of an electron ejected from a piece of sodium (Φ = 4.41x10–19 J) that is illuminated with 265 nm light. in Joules...
- C Chemistry Which pair of compounds has the same empirical formula? A) C2H6 and CH B) CH3 and C2H6 C) C3H8 and C3H D) C2H2 and C2H...
- C Chemistry What is a fossil? The remains of an animal or plant preserved within a layer of rock? A orange body of compacted ice formed over thousands of years? The energy radiated from...
- C Chemistry A student reacts 13 moles of iron with 21 moles of oxygen according to the following equation: 4Fe(s) + 3O2(g) +2Fe2O3(s) How many moles of iron(III) oxide will the reaction...
- C Chemistry How many moles are in 5,5 x 1023 molecules of H2O...
- C Chemistry How many moles of KBr will be produced from 7.0 moles of BaBr2?...
- C Chemistry Pleeeasseee help 32.5 grams of NaOH reacts with an excess of CO2 2NaOH(s) + CO2(g) ⇒ Na2CO3(s) + H20 What is the theoretical yield of Na2CO3 in grams? *** I NEED YOU TO SHOW...
- C Chemistry Briefly describe how plasma TVs display colour....
- C Chemistry 10. Compare the size of an atom to the size of the ion that it forms....

Ответ:
The given question is incomplete. The complete question is as follows.
A solution of chloroform (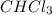 ) and acetone (
) and acetone (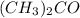 ) exhibits a negative deviation from Raoult's law. This result implies that:
) exhibits a negative deviation from Raoult's law. This result implies that:
chloroform-chloroform interactions are stronger than chloroform-acetone interactions.
chloroform-chloroform interactions are weaker than chloroform-acetone interactions.
acetone-acetone interactions are stronger than chloroform-acetone interactions.
acetone-acetone interactions are weaker than chloroform-acetone interactions
chloroform-chloroform interactions are weaker than chloroform-acetone interactions AND acetone-acetone interactions are weaker than chloroform-acetone interactions.
Explanation:
It is known that when the forces between the particles of the mixture are stronger than the forces between the particles in the pure liquids then negative deviations from Raoult's law are observed.
Hence, when a solution of chloroform (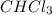 ) and Acetone (
) and Acetone (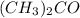 ) will exhibit a negative deviation from Roult's law.
) will exhibit a negative deviation from Roult's law.
Then, chloroform-chloroform interactions should be weaker than chloroform-acetone interactions and acetone-acetone interactions must be weaker than Chloroform-Acetone interactions.
thus, we can conclude that for the given situation chloroform-chloroform interactions are weaker than chloroform-acetone interactions AND acetone-acetone interactions are weaker than chloroform-acetone interactions.
Ответ: