murdocksfamilyouoxvm
28.01.2020 •
Chemistry
Assume that each atom is a sphere, and that the surface of each atom is in contact with its nearest neighbor. determine the percentage of unit cell volume that is occupied in (a) a face- centered cubic lattice, (b) a body-centered cubic lattice, and (c) a diamond lattice.
Solved
Show answers
More tips
- H Health and Medicine What to Take with You to the Maternity Hospital?...
- F Family and Home How to Choose the Perfect Air Conditioner for Your Life...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
Answers on questions: Chemistry
- C Chemistry Consider the following half-reactions and their standard reduction potentials then give the standard line (cell) notation for a voltaic cell built on these half reactions. mn2+(aq)...
- C Chemistry Agalvanic (voltaic) cell consists of an electrode composed of chromium in a 1.0 m chromium(iii) ion solution and another electrode composed of copper in a 1.0 m copper(ii) ion...
- C Chemistry Which of the following statements concerning entropy is/are correct? 1. the entropy of a substance increases when converted from a liquid to a solid. 2. the entropy of a substance...
- C Chemistry What single condition is required to insure that a molecule will be polar covalent? (a) a bond between atoms that are of widely different atomic weights. (b) a bond between atoms...
- C Chemistry How would one convert 6 lb to ounces (16 oz = 1 lb)? A. 6lb 16 oz 1 lb Oo 6 lb 16 oz O B. 1662 O c. 616x 15 O D. 6 lb 16 oz SUBMIT...
- C Chemistry Given the following equation n2o(g) + no2(g) → 3no(g) ∆h =-92 kj/mol A. Is this reaction exothermic or endothermic? B. What direction with the equilibrium shift if Nitrogen gas...
- C Chemistry What is in group 6 period 2 in the periodic table...
- C Chemistry How would I find H3O in this problem using the PH...
- C Chemistry Give four differences between rusting and burning...
- C Chemistry ¿Cuánto se paga por 1 kWh Perú 2020?...

Ответ:
Explanation:
The percentage of unit cell volume = Volume of atoms/Volume of unit cell
Volume of sphere =
a) Percentage of unit cell volume occupied by atoms in face- centered cubic lattice:
let the side of each cube = a
Volume of unit cell = Volume of cube = a³
Radius of atoms =
Volume of each atom = =
= 
Number of atoms/unit cell = 4
Total volume of the atoms =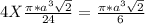
The percentage of unit cell volume =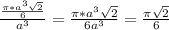 = 0.7405
= 0.7405
= 0.7405 X 100% = 74.05%
b) Percentage of unit cell volume occupied by atoms in a body-centered cubic lattice
Radius of atoms =
Volume of each atom =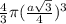 =
=
Number of atoms/unit cell = 2
Total volume of the atoms =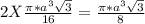
The percentage of unit cell volume = = 0.6803
= 0.6803
= 0.6803 X 100% = 68.03%
c) Percentage of unit cell volume occupied by atoms in a diamond lattice
Radius of atoms =
Volume of each atom =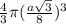 =
= 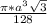
Number of atoms/unit cell = 8
Total volume of the atoms =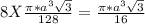
The percentage of unit cell volume = = 0.3401
= 0.3401
= 0.3401 X 100% = 34.01%
Ответ:
A
Explanation: