marelinatalia2000
03.02.2021 •
Chemistry
At STP, a 12,0-liter sample of CH4(g) has the same total number of molecules as
1
6.0 L of H2(g) at STP
2
120 L of CO2(g) at STP
3
18.0 L of HCI(g) at STP
24.0 L of O2(g) at STP
Solved
Show answers
More tips
- T Travel and tourism Lost in the Catacombs: What to Do?...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
Answers on questions: Chemistry
- C Chemistry The number of moles of h+ ions in a liter of distilled (pure) water is: 1 x 10-7 1 x 10-14 0.5 x 10-7 2 x 10-14 none of these...
- B Biology HHRRTSEE rearrange this...
- G Geography What has been the main force behind taiwan’s economic growth? agri-business subsistence farming manufacturing nuclear weapons...

Ответ:
-
In the acetylene torch, 7.5 moles of O₂ are needed to react with 3.00 moles of acetylene gas C₂H₂.
Explanation: -
The balanced chemical equation for this reaction is
2 C₂H₂ + 5 O₂ → 4 CO₂ + 2 H₂O
From the equation we see
2 moles of C₂H₂ react with 5 moles of O₂
3.00 moles of C₂H₂ react with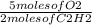 x 3.00 moles of C₂H₂
x 3.00 moles of C₂H₂
= 7.5 moles of O₂
Thus, in the acetylene torch, 7.5 moles of O₂ are needed to react with 3.00 moles of acetylene gas C₂H₂.