melkumathurin
18.08.2019 •
Chemistry
Aweak acid, hf, is in solution with dissolved sodium fluoride, naf. if hcl is added, which ion will react with the extra hydrogen ions from the hcl to keep the ph from changing?
Solved
Show answers
More tips
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
Answers on questions: Chemistry
- C Chemistry What are some antonyms for filament...
- C Chemistry In four paragraphs write your vision of the future of all life on our planet in regards to food, water, hunger, and the environment as a whole. be creative but realistic....
- C Chemistry The craters on the moon are evidence of what events? o impacts with other objects mountain formations volcanoes on the moon alien life...
- C Chemistry Calculate the number of moles in 56g of h20...
- C Chemistry What is the correct way to represent the ionic compound sodium fluoride?...
- C Chemistry A container of hydrogen gas has the same temperature as a container of oxygen gas. The gas atoms having the greater average kinetic energy are the:...
- C Chemistry How many total electrons can the p orbitals hold?...
- C Chemistry Why is melting point considered a physical property...
- C Chemistry Calculate the equilibrium concentration of Ag+(aq ) in a solution that is initially 0.140 M AgNO3 and 0.890 M NH3, and in which the following reaction takes place: Ag+...
- C Chemistry Why does gold sink and wood float when they have similar volumes?...

Ответ:
Explanation:
The weak acid HF is in solution with dissolved NaF; NaF is an ionic compound, so it will dissolve by dissociating its ions; this solution reaction is:
NaF⇒
So, initially there will be these species in solution:
-NaF
-
-
If we add HCl, which is a strong acid, it will dissociate completely (that is the characteristic of a strong acid) in ions H+ and Cl-:
HCl⇒
So, we are finally adding ions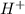 and
and  .
.
We are asked which ion will react with the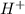 released by HCl acid. We should think just about the anions (negative ions) because we require an electron donor to create a bond with H+, so it won't be on solution as H+. We just have two anions in solution:
released by HCl acid. We should think just about the anions (negative ions) because we require an electron donor to create a bond with H+, so it won't be on solution as H+. We just have two anions in solution:  and
and  .
.
If bonded to
bonded to 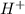 it would form HCl, but we saw that HCl is a strong acid which always dissociate completely in aqueous solution; so
it would form HCl, but we saw that HCl is a strong acid which always dissociate completely in aqueous solution; so  will never bond to
will never bond to 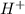 . Finally,
. Finally,  can bond
can bond 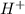 giving HF as a result; it is possible because HF is a weak acid, and HF can be, in fact, present in an aqueous solution; the
giving HF as a result; it is possible because HF is a weak acid, and HF can be, in fact, present in an aqueous solution; the 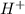 ions will be changed to HF and the pH (which depends only on
ions will be changed to HF and the pH (which depends only on 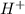 concentration) will not change.
concentration) will not change.
Ответ:
https://quizlet.com/4424118/501-502-503-504-flashcards/