Bamaboy8804
03.12.2020 •
Chemistry
Balance the following equations:
i. H2SO4 + ….NaHCO3 → Na2SO4 + ….H2O + ……CO2
ii. H2 + I2 → ….HI
iii. …..NaOH + H2SO4 → Na2SO4 + …..H2O
iv. ……FeSO4 → Fe2O3 + SO2 + SO3
v. C3H6O2 + …..O2 → ….CO2 + …..H2O
b) Calculate the molecular masses of the following:
i. H2SO4
ii. CH4
iii. NH3
c) 56g of iron reacts with 71g of chlorine. How many grams of iron react with 35.5g of chlorine?
Solved
Show answers
More tips
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...

Ответ:
(I)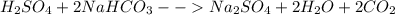
(II)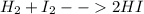
(III)
(IV)
(V)
_____________________________________Molecular Masses of the following:------------------------------------------------------------------------------------------------------------
(I)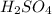
(ANS)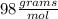
__________________________________________________________
Solution:
Molecular mass of,
H = 1
S = 32
O = 16
Now,
There are 2 atoms of Hydrogen, 1 atoms of Sulfur and 4 atoms of Oxygen present, thus,

_____________________________________(II)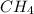
(ANS)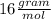
_________________________________________________________
Solution:
Molecular Mass,
Carbon = 12
Hydrogen = 1
Now
There are 1 atom of Oxygen and 4 atoms of Hydrogen present, thus,
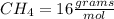
_____________________________________(III)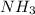
(ANS)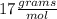
__________________________________________________________
Solution:
Molecular Mass,
Nitrogen = 14 grams
Hydrogen = 1
Now,
There are 1 atoms of Nitrogen and 3 atoms of Hydrogen present, thus,
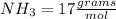
_____________________________________How many Grams of Iron(fe):56 71
x 35.5
_____________________________________Simplify the equation
X = 28 grams
__________________________________________________________
28 grams of Iron(fe) will react with chlorine.
_____________________________________Best Regards,'Borz'Ответ:
A. Four C–H single bonds and two O=O double bonds.
Explanation:
The chemical reaction proceeds with breaking bonds in the reacting molecules and forming new bonds in the products.In this reaction four single bonds are broken in CH₄ and two double bonds O=O are broken in two molecules of O₂.Also, two double bonds C=O are formed in CO₂ molecules and 4 single bonds O–H are formed in 2 molecules of H₂O (each molecule contain 2 new bonds).So, A is the right answer.