Be sure to answer all parts. Butane gas is compressed and used as a liquid fuel in disposable cigarette lighters and lightweight camping stoves. Suppose a lighter contains 7.96 mL of butane (d = 0.579 g/mL). (a) How many grams of oxygen are needed to burn the butane completely?
Solved
Show answers
More tips
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- H History Who was the supreme allied commander during world war 2 douglas macarthur george c. marshall chester nimitz dwight eisenhower...
- P Physics What happens in this circuit if one of the light bulbs is unscrewed?...
- C Chemistry Why do particles in a matter vibrate/move?...
- S Social Studies When a political group grandstands or exaggerates its position to the public, it can be said it is...
- H Health Scientists enroll 500 healthy adults in a study and collect dietary and other lifestyle information about the group. After 6 years, the scientists determine that study participants...

Ответ:
The correct answer will be "4.60 g".
Explanation:
The given values are:
Volume of Butane = 7.96 mL
Density = 0.579 g/mL
As we know,
⇒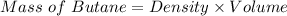
On putting the estimated values, we get
⇒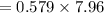
⇒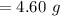
Ответ: