Be sure to answer all parts. Three 8−L flasks, fixed with pressure gauges and small valves, each contain 4 g of gas at 276 K. Flask A contains He, flask B contains CH4, and flask C contains H2. Rank the flask contents in terms of:
Solved
Show answers
More tips
- H Health and Medicine How to Deal with Heat Stroke?...
- F Food and Cooking Discover the Health Benefits of Cedar Nuts...
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- S Science and Technology Colliders: How They Work and Why They Matter...
- A Animals and plants Unraveling the Mystery of Loch Ness: What Does the Loch Ness Monster Look Like?...
- L Leisure and Entertainment How Many Seasons are There in the TV Show Interns?...
- S Sport Playing Bowling: Rules and Advice for Novices...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- C Computers and Internet What is Web 2.0 and How Does it Work?...
Answers on questions: Chemistry
- C Chemistry An object sitting on the floor can have potential energy true or false...
- C Chemistry What is the oxidation number of central metal ion? (b) draw a crystal field splitting diagram of [cr(cn) how many unpaired electrons will there be in central metal ion? (c) write...
- C Chemistry Asample of solid sodium hydroxide (naoh) is added to water at 100°c. as the naoh dissolves, the water boils more rapidly. is the dissolving of naoh in water endothermic or exothermic?...
- C Chemistry (LO 37,45) A reaction is 75% complete in 45.0 min. How long after its start will the reaction be 50% complete if it is (A) first order; (B) zero order...
- C Chemistry A 1.00 g sample of naphthalene (C10H8) contains 0.562 g of hydrogen. What is the mass percent of carbon in the compound?...
- C Chemistry Which solution is neutral? A) Milk of Magnesia B) pure water C) baking soda D) black coffee...
- C Chemistry 3. When a gas is heated, (1 point)...
- M Mathematics If f(x)=2(x)^2+5sqrt(x+2), complete the follwoing statement ( round your answer to the nearest hundreth) : f(0)=...
- H History Many cuban americans fled the dictatorship of fidel castro and no reside in south florida. true or false?...
- A Arts Newsreels showed only recreations of newsworthy events. true or false?...

Ответ:
Here is the complete question.
Be sure to answer all parts. Three 8−L flasks, fixed with pressure gauges and small valves, each contain 4 g of gas at 276 K. Flask A contains He, flask B contains CH4, and flask C contains H2. Rank the flask contents in terms of: the following properties. (Use the notation >, <, or =, for example B=C>A.)
(a) pressure
(b) average molecular kinetic energy
(c) diffusion rate after the valve is opened
(d) total kinetic energy of the molecules
Explanation:
Given that:
Three flask A,B, C:
contains a volume of 8-L
mass m = 4g &;
Temperature = 276 K
Flask A = He
Flask B = H₂
Flask C = CH₄
a) From the ideal gas equation:
PV = nRT
where;
n = number of moles = mass (m)/molar mass (mm)
Then:
PV = m/mm RT
If T ,m and V are constant for the three flasks ; then
P ∝ 1/mm
As such ; the smaller the molar mass the larger the pressure.
Now; since the molecular weight of CH₄ is greater than He and H₂ and also between He and H₂, He has an higher molecular weight .
Then the order of pressure in the flask is :
where :
b)
average molecular kinetic energy
We all know that the average molecular kinetic energy varies directly proportional to the temperature.
Thus; the given temperature = 276 K
∴ The order of the average molecular kinetic energy is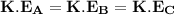
c)
The rate of diffusion of gas is inversely proportional to the square root of it density . Here the density is given in relation to their molar mass.
So;
rate of diffusion ∝
where;
Thus; the order of the rate of diffusion =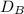 >
>  >
> 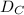
d) total kinetic energy of the molecules .
The kinetic energy deals with how the speed of particles of a substance determines how fast the substances will diffuse in a given set of condition.
The the order of the total kinetic energy depends on the molecular speed
Thus; the order of the total kinetic energy for the three flask is as follows:
Ответ:
B
Explanation:
The carriage has potential energy