alanflores40
29.06.2019 •
Chemistry
Both stearic acid and sucrose are molecular solids, but one is polar and the other is non polar. compare the solubility of the two compounds in water and in hexane to determine which is which
Solved
Show answers
More tips
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- F Family and Home What is Most Important in Men s Lives?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry What is the net ionic equation for the reaction below?...
- E English Passage:His heart beat faster and faster as Daisy s white face came up to his own. He knew that when hekissed this girl, and forever wed his unutterable visions to her perishable...
- H History 1. is the force exerted on the artery walls by the blood flowing through the body. muscle pressure sinus pressure blood pressure respiratory pressure 2. scientific and technological...
- C Chemistry A student claims that she cannot identify the exact location of an electron. Which of the following explains if the student s claim is correct or incorrect? (1 point) The claim...
- E English Which best states how information presented in team moon and the nasa article are different? 1.the nasa article is about the apollo 11 mission, while the team moon article is mostly...
- H History In which form of government do the citizens make the decisions for the government?...

Ответ:
Explanation:
Polar molecule are soluble in polar solvents Non polar molecules are soluble in non polar solvents.Stearic acid is an organic acid composed of seventeen carbons with one carboxylic acid group in the end. Its chemical formula is . The hydrophobic part is this chain is made up of 17 carbon atom and hydrophilic part is only of made up of carboxylic group due to which it won't get dissolve in water.Which means that stearic acid will get dissolve in non polar solvent that is hexane. Hence, stearic acid is non polar molecule.
. The hydrophobic part is this chain is made up of 17 carbon atom and hydrophilic part is only of made up of carboxylic group due to which it won't get dissolve in water.Which means that stearic acid will get dissolve in non polar solvent that is hexane. Hence, stearic acid is non polar molecule.
Where as sucrose an organic molecule with molecular formula of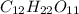 .Due to presence of polar hydroxyl(-OH) groups in molecule , molecule will get easily dissolve in polar solvent that is water. Since,the hydrophobic part in the molecules are covered by the hydrophilic part due to which it gets easily dissolve in water. Hence, the sucrose in polar molecule.
.Due to presence of polar hydroxyl(-OH) groups in molecule , molecule will get easily dissolve in polar solvent that is water. Since,the hydrophobic part in the molecules are covered by the hydrophilic part due to which it gets easily dissolve in water. Hence, the sucrose in polar molecule.
Ответ:
i do what do you need help with
Explanation: