C3H8 + 5O2 —> 3CO2 + 4H2O
How many grams of oxygen are needed to burn 5.0 moles of C3H8?
Please answer
Solved
Show answers
More tips
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- B Biology Which Punnett square represents a cross between two parents that are heterozygous for purple flowers? F f FF Ff f F ff f f Submit Nit...
- E English What is the comprehensive definition for justification (relating to argumentation)...
- P Physics EXERCISE 1 An object and an image are displayed on either side of a lens. Copy the figure to your sheet. A) Calculate the focal length using the lenses formula. B) Determine...
- H History Which of these states was not one of the original Thirteen Colonies? Ner HanapshireMarylandVersoGeorgia...

Ответ:
The correct answer will be "4.60 g".
Explanation:
The given values are:
Volume of Butane = 7.96 mL
Density = 0.579 g/mL
As we know,
⇒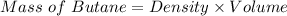
On putting the estimated values, we get
⇒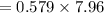
⇒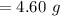
Ответ:
it spreads out through whatever container it is in
Explanation: